Form IF Rubiscos include highly active, specific, and small subunit-independent enzymes.
Form IF Rubiscos include highly active, specific, and small subunit-independent enzymes.
Otto, F.; Westedt, H.; Franzeck, K. P.; Zarzycki, J.; Kueffner, A. M.; Schulz, L.; Prinz, S.; Paczia, N.; Claus, P.; Hochberg, G. A. K.; Erb, T. J.
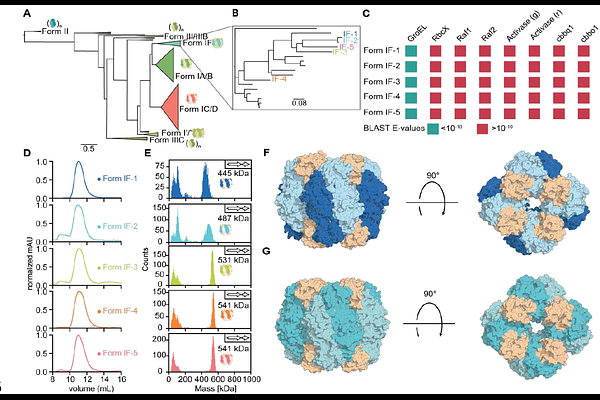
AbstractPlant-type (Form I) Ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) suffers from inherent catalytic trade-offs and a strong dependency on other proteins--including an essential small subunit (SSU) and auxiliary chaperones--for assembly, constraining the enzyme's evolutionary and engineering potential. Here, we investigated representatives from the newly discovered clade Form IF. These enzymes do not require specific chaperones to form functional complexes, exhibit high CO2 -specificities (SC/O ~50) while maintaining high turnover rates (up to kcat ~11 s-1). Remarkably, two Form IF representatives (IF-1/IF-2) lost the dependency on the SSU and assemble into homo-octameric complexes without their cognate SSUs. While the SSU is not necessary for catalysis, its addition improves both activity and specificity in IF-1/IF-2. Our results show that complexity is actually not required to achieve highly active, specific and functional Rubisco variants--and that this complexity can even be reverted--which challenges our current thinking on the evolution and catalytic mechanism of Rubisco.