Cellular thermal shift assay of subcellular isolates for evaluating drug-membrane target interactions
Cellular thermal shift assay of subcellular isolates for evaluating drug-membrane target interactions
Dogra, S. K.; Kattunga, V.; Mookerjee, S.; Rane, A.; Chamoli, M.; Andersen, J.
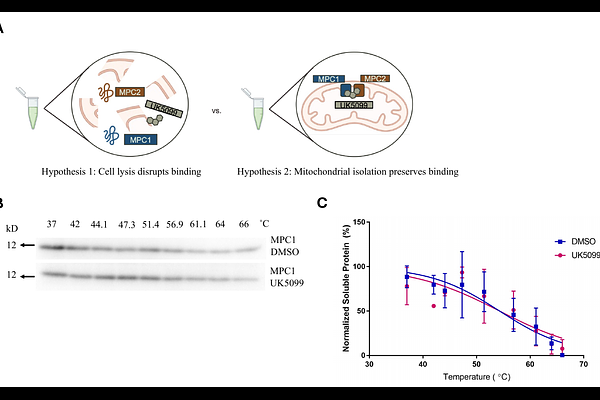
AbstractThe cellular thermal shift assay (CETSA) is an invaluable tool for target identification and validation in early drug discovery efforts. It relies on thermal melting curves to indicate drug binding and is typically performed in whole cells, cell lysates, or purified protein as validation of direct interaction. However, these approaches can result in disruption of the structural integrity of membrane proteins, hindering downstream analysis and drug-target engagement. Here, we describe the first application of CETSA in isolated mitochondria and show the effects of this approach on the analysis of the compound UK5099 and its known binding target, the mitochondrial pyruvate carrier (MPC), a mitochondrial inner membrane localized protein complex. Our analysis supports a model in which the MPC must remain structurally intact for UK5099 binding. We demonstrate that the binding of UK5099 to the MPC is disrupted in whole cells and cell lysates, whereas isolating mitochondria maintains the binding interaction between drug and target observable using CETSA. These data suggest that isolating membrane-bound organelles through subcellular CETSA stabilizes membrane-bound proteins in their native conformation, allowing the identification of membrane-localized drug binding targets that might otherwise be missed.