Structural Basis for C8 methylation of 23S ribosomal RNA by Cfr
Structural Basis for C8 methylation of 23S ribosomal RNA by Cfr
Esakova, O. A.; Jung, J.; Lee, H.; Cho, S. H.; Alumasa, J. N.; Schwalm, E.; Grove, T. L.; Bauerle, M.; Hafenstein, S.; Yu, Z.; Booker, S.
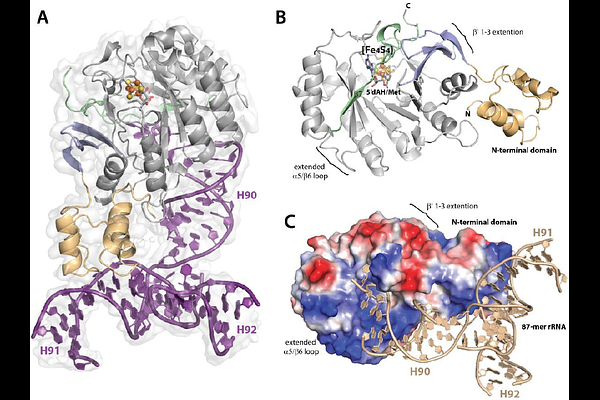
AbstractCfr methylates C8 of adenosine 2503 (A2503) in 23S ribosomal RNA (rRNA) and will also methylate C2 of A2503 after methylating C8. C8 methylation confers resistance to more than five classes of clinically used antibiotics, highlighting it as a worrisome mechanism of antibiotic resistance. Here, we report the structure of Cfr, determined by cryogenic electron microscopy (Cryo-EM). Despite its small size (~36 kDa), we exploit a transient protein-RNA crosslink that forms during catalysis, which requires Cys105 to resolve. Using a Cfr Cys105Ala variant and an 87-nucleotide strand of rRNA, we isolate the crosslinked species and determine its structure to 3.0 Angstroms resolution. Notably, the 87-mer rRNA adopts an L-shaped conformation characteristic of tRNAs, rather than the conformation it assumes in the ribosome.