Multicenter preclinical validation of next-generation CAR T cells: a strategy for harmonization, reproducibility, and its feasibility in clinical translation
Multicenter preclinical validation of next-generation CAR T cells: a strategy for harmonization, reproducibility, and its feasibility in clinical translation
Dalloul, I.; Barden, M.; Wilcke, J.; Bernhard, S.; Ellenbach, N.; Boulesteix, A.-L.; Abken, H.; Kobold, S.
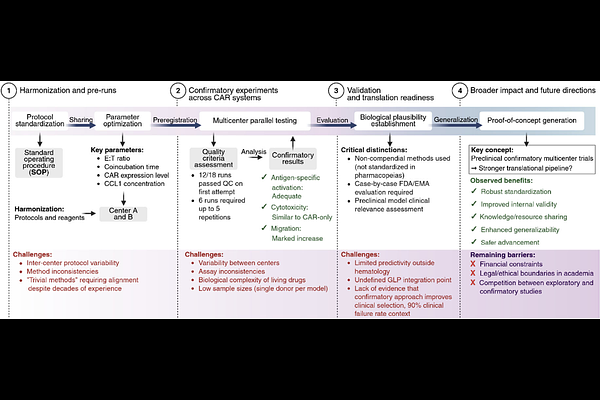
AbstractPurpose: Clinical translation of CAR T cell therapies has accelerated, yet preclinical evidence still often originates from single-center studies lacking sufficient robustness. Preclinical confirmatory multicenter studies have been proposed to improve the translational success, but their feasibility in cellular therapies remains unexplored. Methods: We performed a confirmatory multicenter study validating C-C-motive-receptor-8 (CCR8) overexpression in CAR T cells; a strategy previously shown to enhance solid tumor infiltration. In vitro experiments covering activation, cytotoxicity, and migration using three CAR constructs were conducted across two centers with harmonized materials, preregistered protocols, randomization, and blinding. Results: The data from the two centers confirmed key findings of the exploratory study: CCR8 overexpression in anti-EpCAM and anti-mesothelin CAR T cells leads to enhanced selective migration towards a CCL1-gradient, while not compromising antigen-specific T cell activatory capacity and cytotoxicity in vitro. The study furthermore broadened the applicability of CCR8 overexpression to anti-CEA CAR T cells. Conclusions: This first-of-its-kind preclinical confirmatory CAR T study demonstrates the feasibility of a multicenter confirmation in cellular therapy, with technical and logistical challenges resolved through transparent communication between all parties involved. Both exploratory and confirmatory studies aim to downselect CAR candidates with the highest clinical success potential, as they compete for limited resources in preclinical research. It is therefore mandatory to clarify the extent of replications required to validate the experimental methodology and identify CAR candidates with most likelihood of success.