Molecular genetic characterization of bacterial KH-domain proteins
Molecular genetic characterization of bacterial KH-domain proteins
Nguyen, K. T.; Lett, N. W.; Gravel, C. M.; Jo, S.; Shi, Y.; Narayan, M.; Sharma, S.; Sharma, C. M.; Berry, K. E.
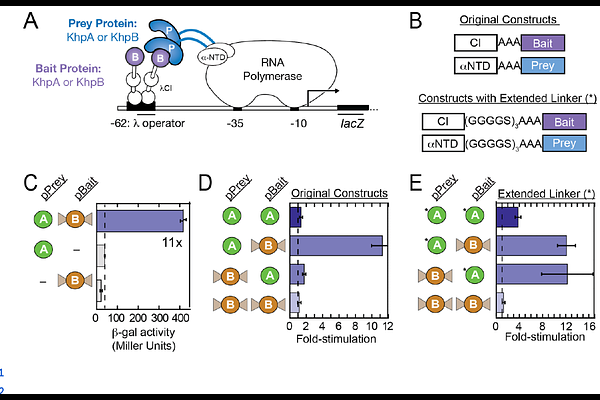
AbstractPost-transcriptional gene regulation is a key mechanism for bacterial stress responses and virulence, and RNA-mediated regulation frequently relies on global RNA-binding proteins (RBPs). A pair of interacting KH-domain proteins (KhpA and KhpB) have recently been identified as global RBPs in several bacterial species. To better understand their molecular functions, we employed bacterial two- and three-hybrid (B2H/B3H) assays in an E. coli reporter system to analyze protein-protein and protein-RNA interactions of KhpA/B orthologs from three human pathogens: Campylobacter jejuni, Helicobacter pylori, and Clostridioides difficile. Protein-protein interactions were conserved across all species, with KhpA-KhpB heterodimers forming more robustly than either homodimer and KhpA homodimerizing more readily than KhpB. On the other hand, protein-RNA interactions were more varied across species: C. jejuni and C. difficile KhpA bound both species-specific and non-specific RNAs, but H. pylori KhpA--and KhpB orthologs from all species--showed no RNA interaction in B3H assays. Site-directed mutagenesis experiments demonstrated that residues in the GXXG motif of KhpA are critical for RNA interaction and differences in these residues account for the distinct RNA-binding behaviors of KhpA orthologs. Collectively, these findings provide a cross-species, molecular view of how KhpA and KhpB recognize one another and RNA ligands to regulate gene expression.