Cryo-EM structures of photosystem I with alternative quinones reveals new insight into cofactor selectivity
Cryo-EM structures of photosystem I with alternative quinones reveals new insight into cofactor selectivity
Brininger, C. M.; Wang, J.; Kurashov, V.; Russell, B. P.; Magdaong, N. C. M.; Iwig, D. F.; Est, A. v. d.; Golbeck, J. H.; Vinyard, D. J.; Lakshmi, K. V.; Gisriel, C. J.
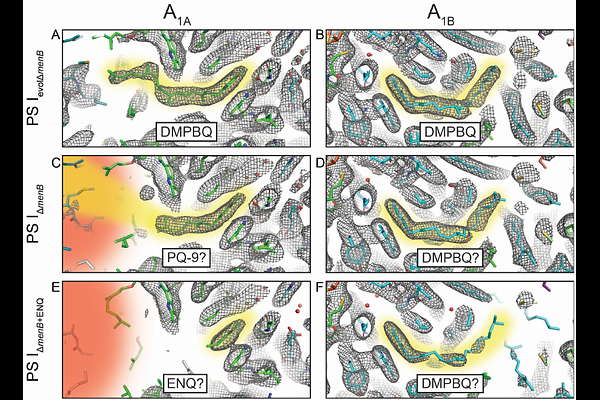
AbstractQuinones are an integral component of electron transfer processes in photosynthetic and mitochondrial respiratory proteins. One such photosynthetic protein, Photosystem I, is an essential photooxidoreductase found in all oxygenic phototrophs. To better understand quinone chemistry and to form a basis for protein engineering, the menB gene in the model cyanobacterium Synechocystis sp. PCC 6803 was interrupted, blocking the biosynthesis of phylloquinone and causing it to be replaced by exchangeable plastoquinone-9 in the A1A and A1B quinone-binding sites of Photosystem I. This genetic variant has been instrumental in bioenergy research, enabling incorporation of a range of substituted and isotopically labeled quinones. Despite numerous valuable studies, the interpretation of biophysical data has been limited by a lack of structural data. To address this, we present the high-resolution cryo-EM structures of Photosystem I from the {Delta}menB variant containing (a) exchangeable plastoquinone-9 and (b) exogenously added 2-ethyl-1,4-napthoquinone at 1.90- and 2.05-A resolution, respectively. Unexpectedly, the quinones in the A1A and A1B sites of Photosystem I, previously believed to have similar binding affinities, are found to be asymmetric in their ability to bind and exchange plastoquinone-9. This work reveals new and important insight into the molecular basis for Photosystem I activity in the {Delta}menB variant, the power of metabolic plasticity to maintain protein stability, and the requirement for protein instability to facilitate ligand exchange.