Early Epigenetic and Metabolic Responses to the Adipocyte Secretome Reveal Stress-Adaptive States in Triple-Negative Breast Cancer
Early Epigenetic and Metabolic Responses to the Adipocyte Secretome Reveal Stress-Adaptive States in Triple-Negative Breast Cancer
Townsel, A.; Jaffe, M.; He, S.; Wu, Y.; Ingram, A.; Tipton, M.; Kemp, M. L.; Henry, C. J.; Haynes, K. A.
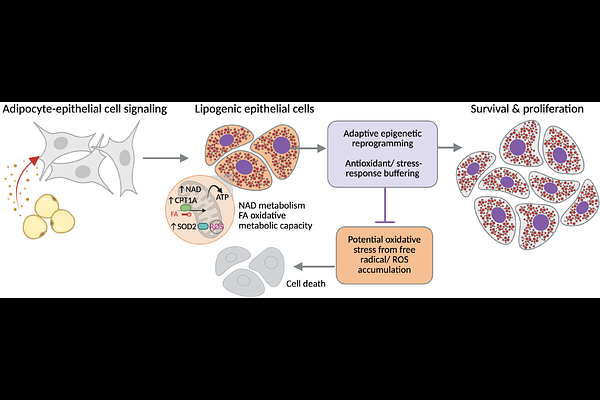
AbstractObesity is a well-established risk factor for triple-negative breast cancer (TNBC), yet how adipocyte-derived signals reprogram cancer cell metabolism and chromatin states remains poorly defined. Here, we investigate how adipocyte-driven lipogenesis reshapes metabolic-epigenetic coupling to support stress-adaptive cell states and functional changes in epithelial TNBC cells. Using an integrated multi-omic approach, we combine RNA sequencing (RNA-seq), chromatin accessibility (ATAC-seq), metabolic flux modeling, and functional metabolic assays in lipogenic BT-549 cells. Computational modeling trained on RNA-seq predicts shifts in metabolic pathway usage, including enhanced NAD-linked metabolism. RNA-seq reveals a predominance of gene activation, consistent with ATAC-seq data showing a strong bias toward increased accessibility. Regions of increased accessibility are enriched for stress-adaptive and antioxidant pathways, including superoxide dismutase 2 (SOD2) and metallothioneins (MT1F, MT1E, MT2A). Functionally, lipogenic cells exhibit increased spare respiratory capacity, altered ATP-linked respiration, elevated extracellular acidification, and reduced reactive oxygen species (ROS) accumulation, consistent with a bioenergetically flexible, stress-adaptive metabolic state. Together, these findings reveal that adipocyte-driven metabolic rewiring promotes selective chromatin opening and activation of stress-adaptive gene programs, enabling TNBC cells to buffer oxidative pressure for enhanced proliferation and survival after exposure to the adipocyte secretome.