Fibroblast-derived Collagen VI shapes the structure and function of the tumor-immune microenvironment in clear cell renal cell carcinoma
Fibroblast-derived Collagen VI shapes the structure and function of the tumor-immune microenvironment in clear cell renal cell carcinoma
Wess, M.; Andreev, G.; Feilen, T.; Diel, N.; Zirngibl, M.; Gueib-Picard, C.; Koessinger, A. L.; Hinrichs, C.; Naegel, A.; Essmann, C. L.; Roemer, W.; Helmstaedter, M.; Vollmer, T.; Werner, M.; Grabbert, M.; Schilling, O.; Rogg, M.; Schell, C.
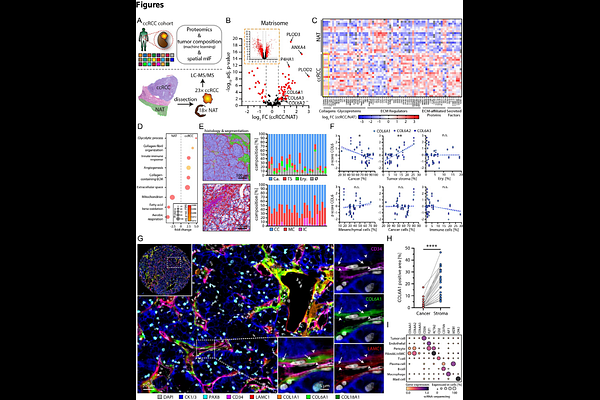
AbstractThe importance of the extracellular matrix (ECM) influencing tumor biology in stroma-rich tumors is well established. However, the relevance of individual ECM proteins in rather stroma-poor cancers such as clear cell renal cell carcinoma (ccRCC) is ill-defined. Using bulk proteomics, spatial imaging, and single-cell transcriptomics, we identify collagen VI (COL6) as a predominant ECM component of the ccRCC interstitial stroma, synthesized primarily by fibroblasts and pericytes. Using cell-derived matrix (CDM) models, we demonstrate that COL6 is essential for maintaining an isotropic ECM network architecture and governs the broader matrisomal composition, with direct pro-proliferative consequences for tumor cells both in vitro and in situ. Granular spatial analysis reveals that COL6-rich stromal septa constrain tumor-infiltrating T cells to boundary zones, where CD8+PD1+ phenotypes predominate. Importantly, tyrosine kinase inhibition (TKI) with cabozantinib suppresses COL6 expression in fibroblasts in vitro and in ex vivo tumor models, mirroring COL6-depleted CDM phenotypes. Our findings establish COL6 as a central stromal regulator of ccRCC tumor biology and immune contexture, revealing ECM remodeling as an underappreciated mechanism of TKI action, with implications for combination immunotherapy strategies.