A stapled peptide inhibitor of MDM2 enables pharmacological activation of p53 in zebrafish
A stapled peptide inhibitor of MDM2 enables pharmacological activation of p53 in zebrafish
Kheder, S.; Krkoska, M.; Mihalic, F.; Kobar, K.; Andrysik, Z.; Bräutigam, L.; Lindström, S.; Berman, J. N.; Lane, D. P.; Lama, D.; Kannan, P.
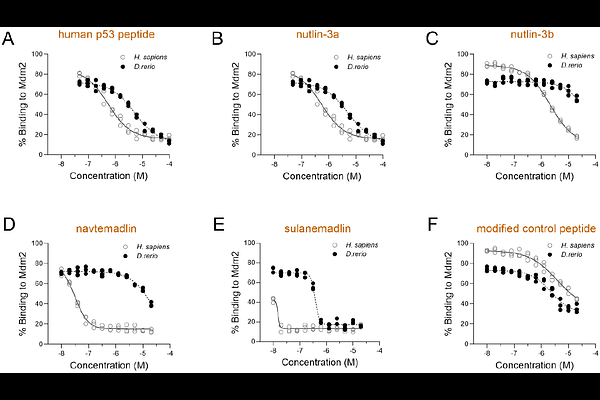
AbstractMeasuring the activity of the tumor suppressor p53 in living systems is essential for understanding its dysregulation in cancer and other conditions, such as aging and diabetes. Zebrafish (Danio rerio) are a powerful vertebrate model that enable such studies, due to the evolutionary conservation of p53 structure and function. However, p53 activity in zebrafish has mainly been assessed using pharmacological methods that induce DNA damage or have off-target effects, making it difficult to isolate p53-specific responses from broader stress responses. Here, by using biophysical assays, molecular dynamics, and molecular assays, we show that sulanemadlin, a stapled peptide inhibitor of MDM2, binds to zebrafish Mdm2 and transcriptionally activates downstream targets of p53, including cdkn1a, isoform {Delta}113p53, and Mdm2. No effect on gene expression was observed in embryos treated with a point-modified control peptide or in embryos carrying a mutation that renders p53 transcriptionally inactive. RNA sequencing further confirmed upregulation of p53 signaling and downregulation of DNA replication pathways, while an acridine orange assay showed no detectable increases in apoptosis. In contrast, the tested small molecule Mdm2 inhibitors exhibit reduced binding affinity to zebrafish Mdm2 due to an amino acid variation in the zebrafish Mdm2 binding pocket. By overcoming a species-specific barrier in p53-MDM2 binding, the stapled peptide sulanemadlin is the first pharmacological tool to specifically activate p53 in zebrafish without inducing measurable apoptosis, enabling direct in vivo studies of p53 regulation in cancer and other disease contexts.