Longitudinal in vivo human wound healing model defines key role for smooth muscle cells in ECM remodeling
Longitudinal in vivo human wound healing model defines key role for smooth muscle cells in ECM remodeling
Emmerich, K.; Suri, R.; Yang, D.; Liu, D.; Huffstutler, R.; Dmitrieva, N. I.; Cudrici, C. D.; Schwartzbeck, R.; Ferrante, E. A.; Hsu, I.; Kinoshita, M.; Goel, S.; Dalgard, C.; Nagao, K.; Pinto, A. R.; Boehm, M.; Harper, R. L.
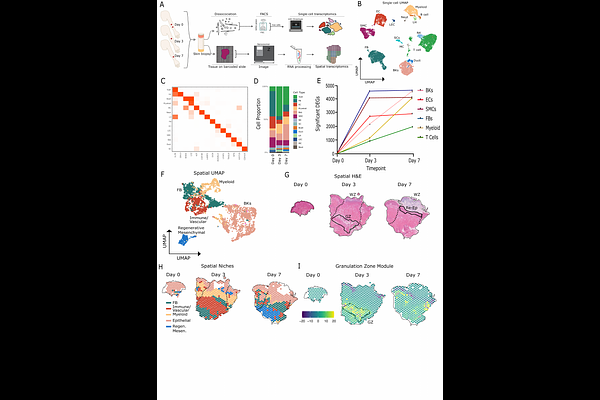
AbstractBackground: Effective skin wound healing is essential for restoring tissue integrity following injury. Repair proceeds through phases of hemostasis, inflammation, proliferation, and remodeling, but molecular mechanisms governing these stages remain poorly defined. Vascular niche cells (VNCs)-including endothelial cells, vascular smooth muscle cells (SMCs), and fibroblasts-are central regulators of healing, but the lack of longitudinal in vivo human data has limited identification of VNC-derived signals that distinguish effective repair from pathological healing such as ulcers. Thus, defining the regulation of VNCs in wound healing addresses a critical knowledge gap. Methods: We developed a protocol for wound healing using dermal forearm punch biopsies to track longitudinal repair in healthy volunteers. Single-cell and spatial transcriptomics were performed to identify and validate signaling activities within VNCs. Results: We spatiotemporally defined the inflammation, proliferation, and remodeling phases of human skin wound healing with a focus on VNCs. Spatial analysis localized this activity for VNCs and immune cells within a heterogenous granulation zone that later led to re-epithelializion. Angiogenesis was dominated by Vegf, Efg and Hif1? signaling. Extracellular matrix (ECM) remodeling occurred through Collagen, Laminin, Thrombospondin, and Fibronectin. SMCs emerged as dominant drivers of injury-induced remodeling including basement membrane and interstitial ECM components compared to fibroblasts. This SMC-led program was further defined by robust induction of TIMP1, an inhibitor of matrix degradation, which localized to granulation tissue and correlated with re-epithelialization and wound resolution. Lastly, we compared remodeling factors between healing and non-healing human diabetic foot ulcers (DFUs). SMCs in non-healing DFUs had deficient expression for core remodeling factors, including TIMP1, indicating SMC activity is needed for effective healing. Conclusion: We identified an SMC-driven model of wound repair in which TIMP1-dependent activity underpins granulation zone formation. Failure of this program defined a mechanistic basis for impaired healing in ulcers, identifying SMCs and TIMP1 as therapeutic targets.