Host outnumbered: microbiomes and a fungal parasite interact to shape host genotype-dependent survival
Host outnumbered: microbiomes and a fungal parasite interact to shape host genotype-dependent survival
Rajarajan, A.; Coone, M.; Wolinska, J.; Spaak, P.; Decaestecker, E.
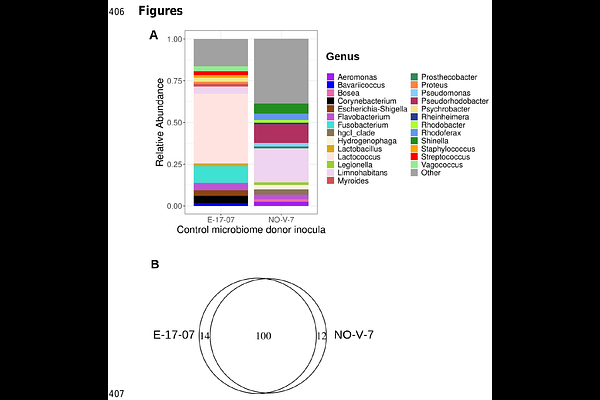
AbstractMicrobiomes are key determinants of host health, yet empirical evidence demonstrat-ing their influence on host-parasite interactions is limited. We conducted a proof-of-principle experiment using the water flea Daphnia magna and its virulent yeast para-site Australozyma monospora (formerly Metschnikowia bicuspidata) to test if microbi-ome composition alters infection outcomes. Axenic hosts were inoculated with a con-trol microbiome (homogenates of laboratory-cultured Daphnia reared in natural fresh-water) or a single bacterial strain (Rhizobium sp.), and exposed to the parasite. Host survival differed markedly between microbiome treatments and depended on parasite exposure. Prior to parasite exposure, hosts of one genotype exhibited high juvenile mortality when inoculated with the control microbiome (79%), compared to Rhizobium (19%) (the other genotype: 48% vs. 50%). Parasite exposure reduced survival, but the extent varied with microbiome composition: survival of hosts with a control micro-biome (averaged across genotypes) declined from 66% to 0%; survival of those inocu-lated with Rhizobium sp. declined from 35% to 10%. In contrast, microbiome composi-tion did not influence parasite infectivity or transmission. Our results indicate that mi-crobiome effects on host survival are genotype- and context-dependent, differing be-tween parasite-free and parasite-exposed conditions. Microbiome composition may therefore impact host-parasite dynamics primarily by influencing host viability rather than parasite performance.