Isosteric Engineering of Enzymes: Overcoming Activity-Stability Trade-offs by Site-Selective CH -> N Substitutions
Isosteric Engineering of Enzymes: Overcoming Activity-Stability Trade-offs by Site-Selective CH -> N Substitutions
Abdelkader, E. H.; Qianzhu, H.; Otting, G.; Huber, T.
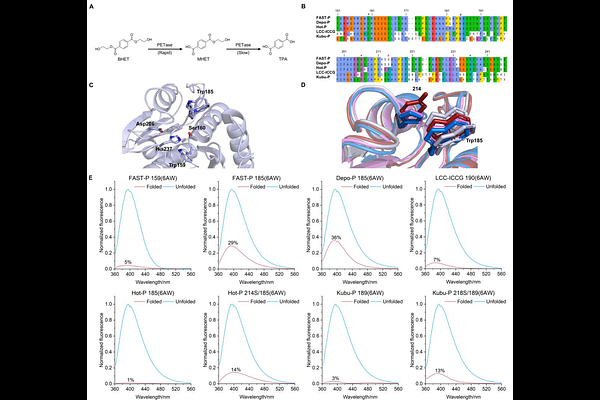
AbstractEnzymes used on industrial scale are routinely engineered for best performance. However, exhaustive mutagenesis campaigns using the twenty canonical proteinogenic amino acids rapidly reach an evolutionary ceiling, where gains in activity compromise other critical properties such as thermal endurance. Although non-canonical amino acids (ncAA) expand the chemical space, most are costly for use on an industrial scale and significantly perturb structure. Here, we demonstrate that the evolutionary ceiling of highly optimized polyethylene terephthalate (PET) hydrolases (PETases) can be broken with azatryptophans that (i) differ minimally from their canonical tryptophan, (ii) are genetically encoded, and (iii) are produced in high yield by enzymatic biosynthesis from inexpensive precursors. The first genetic encoding systems are described for 4-azatryptophan, 5-azatryptophan, and 6-azatryptophan, achieving single, site-selective isosteric CH [->] N substitutions that enhancing the catalytic activity while preserving thermal stability. The fluorescence of 6AW provides a uniquely sensitive reporter of side-chain solvent exposure, which is critical for PETase activity and shown to vary between five different PETases. Furthermore, Azatryptophan-bearing enzymes are inexpensive to produce. To benchmark PETase activity, a rapid fluorescence-based kinetic assay, PETra, is introduced, which delivers consistency and reproducibility by using a soluble substrate yet correlates strongly with the hydrolysis of solid PET.