Purifying selection and phylogenetic discord among microneme proteins in Toxoplasma gondii
Purifying selection and phylogenetic discord among microneme proteins in Toxoplasma gondii
Whittall, J. B.; Zhang, M.; Guiton, P. S.
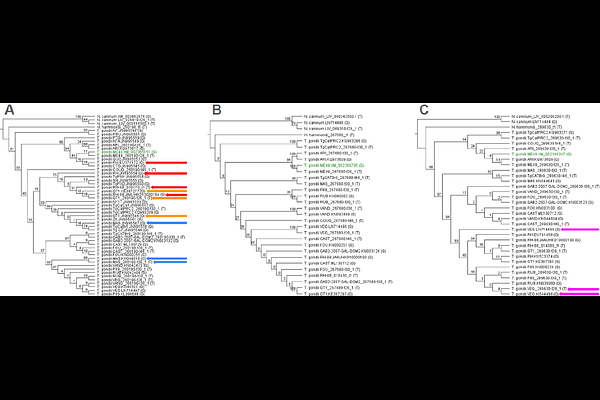
AbstractIn Toxoplasma gondii, microneme proteins (MICs) are secreted components of the apical complex that play central roles in motility, host cell attachment, and invasion. Because proteins at the host-parasite interface are often predicted to evolve rapidly, MICs have been suggested as candidates for adaptive diversification. We tested this expectation using comparative analyses of three relatively understudied microneme proteins, MIC13, MIC12, and MIC16. Coding sequences were assembled from GenBank and ToxoDB, aligned by translation, and analyzed using maximum-likelihood phylogenetics, codon-based tests of selection, and predicted protein structure. MIC13 was represented by 51 sequences, MIC12 by 30, and MIC16 by 34, spanning multiple T. gondii haplogroups and including Hammondia hammondi and Neospora caninum as outgroups. All three genes were highly conserved among T. gondii strains, but their phylogenetic trees were topologically incongruent, indicating that individual MICs do not recover a single shared strain history. Contrary to expectation, no positively selected codons were detected in any gene. Instead, purifying selection was detected at one site in MIC13 and 15 sites in MIC12, while no significant codon-specific selection was detected in MIC16. Several constrained MIC12 sites overlapped annotated EGF and calcium-binding EGF-like domains, consistent with structural conservation of extracellular adhesion modules. AlphaFold prediction of MIC13 supported two sialic acid-binding micronemal adhesive repeat regions, but the single constrained MIC13 site did not overlap these motifs. Together, these results indicate that MIC13, MIC12, and MIC16 are shaped more by sequence conservation and heterogeneous gene histories than by strong recurrent positive selection. These findings refine expectations for microneme evolution in T. gondii and highlight conserved domains that may be important for parasite invasion and future functional study.