Spatially Integrated Multi-Omics reveals the Multicellular Landscape of Progenitor-Driven Glioblastoma Progression
Spatially Integrated Multi-Omics reveals the Multicellular Landscape of Progenitor-Driven Glioblastoma Progression
Traeuble, K.; Traeuble, J.; MOSAIC consortium, ; Kaminski Schierle, G. S.; Heinig, M.
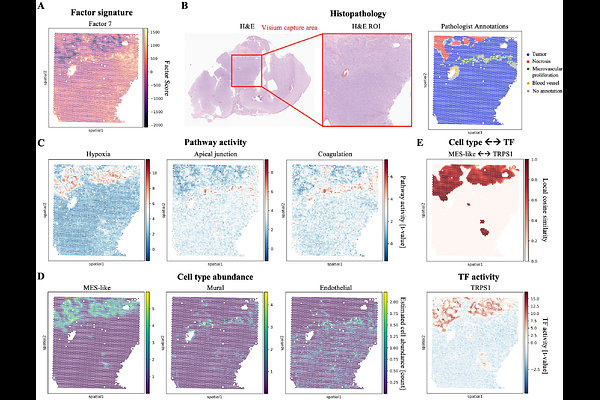
AbstractGlioblastoma is the most lethal primary brain tumor, driven by complex interactions between plastic malignant cells and a diverse tumor microenvironment. Despite advances in single-cell profiling, how genomic drivers and the tumor microenvironment interact to determine tumor progression and patient survival remains poorly understood. While cellular states have been cataloged, the multicellular logic coordinating these into lethal phenotypes remains unresolved. Here, we integrate whole-exome sequencing, bulk and single-cell RNA sequencing, spatial transcriptomics, and histopathology from the MOSAIC cohort (n=89) to deconstruct this inter- and intra-tumoral heterogeneity. We identify a robust latent multi-omic program in glioblastoma that delineates a transition from homeostatic neural precursors to an aggressive, immunosuppressive progenitor phenotype. This high-risk state, which predicts poor survival in both the MOSAIC discovery and independent validation cohorts (TCGA, CGGA; total n=598), is sustained by dense intercellular communication networks linking malignant progenitors with myeloid and endothelial compartments. Spatially, this program maps to hypoxic, perinecrotic niches, directly linking molecular signaling with microvascular proliferation and tissue necrosis. Our findings provide a spatially resolved, multi-omic blueprint of the multicellular logic driving glioblastoma progression, offering a robust molecular framework for patient stratification and targeted intervention.