Discovery of inhibitors of the Pseudomonas aeruginosa NADH:ubiquinone oxidoreductase (NQR) that hinder virulence factors
Discovery of inhibitors of the Pseudomonas aeruginosa NADH:ubiquinone oxidoreductase (NQR) that hinder virulence factors
Ileperuma, S. M.; Li, J.; Ortiz, J.; Shade, T.; Vasseur, C.; Ciancibello, N. A.; Elhenawy, W.; Di Trani, J.
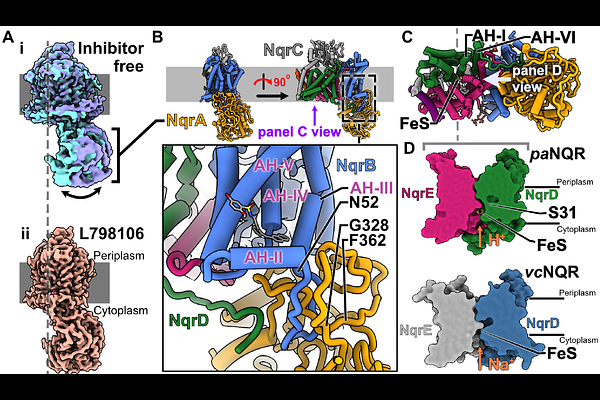
AbstractThe growing threat of antimicrobial resistance has created an urgent need to identify novel therapeutic targets in bacteria. The NADH:ubiquinone oxidoreductase (NQR) is a potential target in a number of bacteria that transfers electrons from NADH to ubiquinone while pumping ions from the cytoplasm to the periplasm. In most species, this complex pumps sodium ions, whereas in Pseudomonas aeruginosa it pumps protons, thereby functioning as a member of the electron transport chain. Using a strain of PAO1 with the NQR knocked out, we demonstrate that the NQR complex plays a crucial role in the motility and biofilm development virulence factors in P. aeruginosa. We develop and execute a high-throughput inhibitor screen to identify and confirm compounds that inhibit NADH oxidation by this complex. Using single-particle cryogenic electron microscopy (cryoEM), we determine high-resolution structures of the NQR complex, both inhibitor-free and bound to one of the confirmed hits from our screen, demonstrating that it binds to the ubiquinone binding site. These structures provide insight into conformational dynamics controlled by binding at the ubiquinone site, with potential implications for the coupling between electron transfer and proton pumping in this complex. Biofilm development and motility assays with selected compounds from the screen show that they affect these virulence factors similarly to the NQR knockout.