Predicting early bactericidal activity of tuberculosis drug combinations using a translational pharmacokinetic-pharmacodynamic modeling approach
Predicting early bactericidal activity of tuberculosis drug combinations using a translational pharmacokinetic-pharmacodynamic modeling approach
de Castro Suarez, N.; Nuermberger, E. L.; Ernest, J.; Savic, R. M.
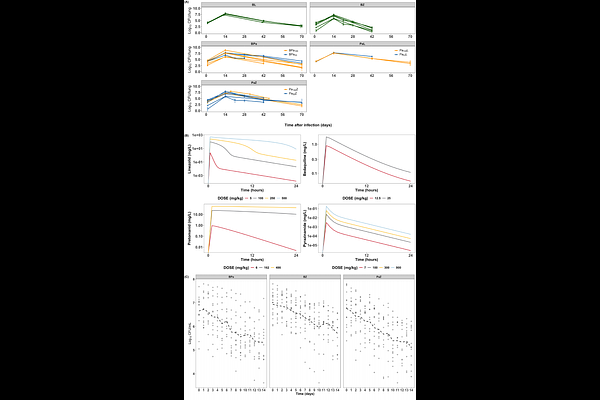
AbstractPhase IIa pulmonary tuberculosis (TB) trials typically assess the early bactericidal activity (EBA) of monotherapy for over 14 days. However, few studies have evaluated drug combinations, even though optimal monotherapy doses may not directly translate to combinations. Translational pharmacokinetic-pharmacodynamic (PK-PD) modeling has shown promise in predicting human treatment responses based on preclinical monotherapy data; however, its application in drug combinations remains limited. This study aimed to extend and validate our previously developed translational monotherapy PK-PD modeling platform to predict the EBA of two-drug combinations. Interactions between bedaquiline, pretomanid, linezolid, and pyrazinamide were characterized using two modeling approaches: the empirical SUPER method and the mechanistic General Pharmacodynamic Interaction model. Both approaches were independently linked to our translational platform and validated using mouse data and Phase IIa clinical results from the NC-001 study. Both modeling methods identified consistent interaction patterns, including antagonistic interactions when bedaquiline was combined with either pretomanid or linezolid. Pyrazinamide has emerged as the most effective companion for both bedaquiline and pretomanid. Our platform reasonably predicted 14-day clinical sputum colony-forming unit counts for multiple two-drug combinations, with most observations falling within the 95% prediction intervals, supporting its use in accelerating regimen development. Our study demonstrated that the translational PK?PD platform reliably predicts both short- and long-term outcomes for combinations, regardless of the interaction model. This supports its application across drug development stages to inform dose selection and effective companion drugs for anti-TB therapies.