Anti-diabetic drug Repaglinide induces Apoptosis, Cell Cycle Arrest, and Inhibits Cell Migration in Human Breast and Lung Cancer Cells.
Anti-diabetic drug Repaglinide induces Apoptosis, Cell Cycle Arrest, and Inhibits Cell Migration in Human Breast and Lung Cancer Cells.
P K, H.; K, A.; Yarla, N. s.; Duddukuri, G. r.
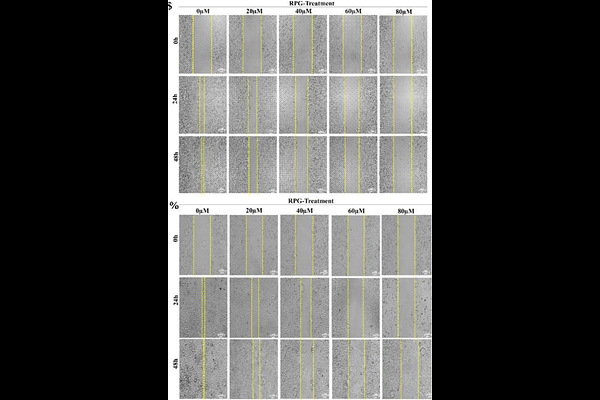
AbstractIntroduction: Drug repurposing offers a cost-effective and time-efficient strategy for cancer therapy by leveraging existing drugs with established safety profiles, thus functioning as an alternative therapeutic strategy in demanding diseases such as cancer. Antidiabetic agents, in particular, have demonstrated encouraging anticancer potential. Among them, the non-sulfonylurea insulin secretagogue repaglinide (RPG) has shown emerging anticancer potential, yet its effects on breast and lung cancers remain largely unexplored. Thus, this study investigates the anticancer activity of repaglinide in human breast (MCF-7) and lung (A549) cancer cell lines, focusing on its cytotoxic, pro-apoptotic, anti-proliferative, and anti-migratory effects and the underlying possible molecular mechanisms. Methodology and Results: MTT cytotoxic assay revealed that RPG reduced cell viability in a dose-/time-dependent manner, with an IC (48h) of 100.8Micromolar for MCF-7 and 104Micromolar for A549. Further, the apoptotic effect of RPG on both cell lines was evidenced by double staining assays, comet assay, and western blotting analysis, suggesting that RPG explicitly caused DNA damage and activated intrinsic and extrinsic apoptosis pathways. Additionally, RPG suppressed clonogenicity and enforced G1 arrest in MCF7 and A549 cells by modulating cell cycle regulations as well as cell proliferation pathways. Moreover, RPG markedly suppressed cell motility, as demonstrated by scratch and Transwell migration/invasion assays, which is correlated with reduced MMP-2 and MMP-9 expression, confirmed by gelatin zymography and western blotting. Conclusion: Conclusively, Repaglinide exerts potent anticancer effects in breast and lung cancer cells by modulating key oncogenic signaling pathways, and thus can be considered a promising candidate for repurposing in cancer therapy.