PARP1 directly disassembles nucleosomes to regulate DNA repair
PARP1 directly disassembles nucleosomes to regulate DNA repair
Verma, A.; Zhu, C.; Truong, B.; Bilokapic, S.; DeVries, R.; Pitre, A.; Shelat, A. A.; Halic, M.; Dao, H. T.
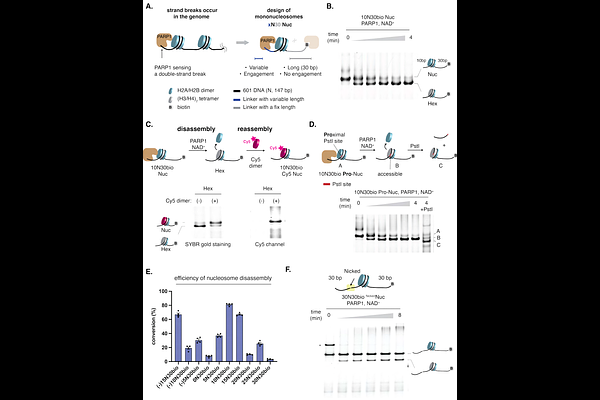
AbstractUpon DNA damage, chromatin remodeling is rapidly initiated to promote chromatin accessibility, thereby facilitating the recruitment and assembly of repair factors. Although this enhanced accessibility has been linked to poly(ADP-ribose) polymerase (PARP) activity, the mechanism by which cells overcome the nucleosome barrier remains unclear. Using our designer chromatin system, we uncovered a previously uncharacterized activity of PARP1, whereby it directly and asymmetrically evicts histone dimers proximal to DNA strand breaks from nucleosomes to generate oriented hexasomes. In the presence of HPF1, PARP1 generates stable PARylated hexasomes, an open chromatin intermediate that can serve as a bifunctional hub for recruitment of DNA- and PAR-dependent factors. Using cellular assays, we demonstrated that PARP activity is both required and sufficient to drive chromatin accessibility and the recruitment of repair factors, with direct involvement of subnucleosomal species. Unexpectedly, we identified the C-terminal tail of histone H2A, a motif harboring recurrent cancer-associated mutations, as a critical determinant of efficient PARP1-mediated nucleosome disassembly. Deletion of the H2A tail sensitizes cells to DNA-damaging agents and PARP inhibitors, implicating a functional role of PARP1-mediated nucleosome disassembly in DNA repair. Together, our findings support a model in which PARP1 directly drives histone eviction, leading to the formation of subnucleosomes that facilitate efficient DNA repair.