Insulin-independent glucose uptake in skeletal muscle by coupled SGLT and Na,K-ATPase transport
Insulin-independent glucose uptake in skeletal muscle by coupled SGLT and Na,K-ATPase transport
Norman, N. J.; Radzyukevich, T. L.; Chomczynski, P. W.; Rymaszewski, M.; Fokt, I.; Priebe, W.; Schmidt, L.; Zhu, T.; Mackenzie, B.; Figueroa, J. L.; Heiny, J. A.
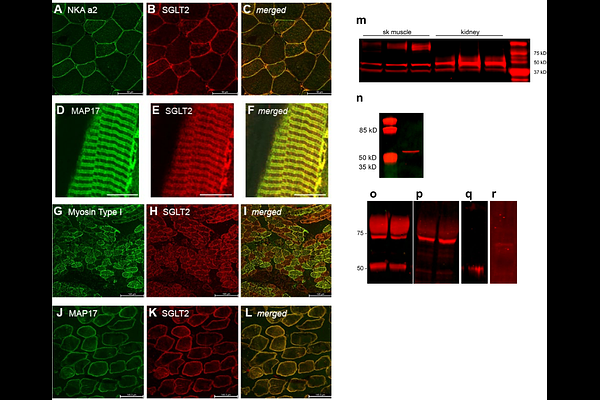
AbstractExercise is a cornerstone therapy for diabetes because working skeletal muscles take up glucose at dramatically greater rates than postprandial insulin-stimulated glucose uptake and, notably, do so without a requirement for insulin. This remarkable ability of working muscles is preserved in diabetes, when muscles become resistant to insulin. However, the mechanism of insulin-independent glucose uptake by working muscles is not fully understood. Here we describe a previously unrecognized glucose uptake pathway in muscle, which we refer to as mSGLT based on shared properties with the Sodium Glucose Linked Transporter family. In contrast to the abundant GLUT4 transporter, mSGLT is not regulated by insulin, requires Na,K-ATPase-2 activity, and transports the hexose alpha-methyl-D-glucoside (-MDG), a glucose derivative that is handled by SGLTs but not GLUT4. The mSGLT pathway and GLUT transport pathways are independent and additive. In addition to exercise, mSGLT imports glucose under other conditions of adrenergic stimulation, which inhibits pancreatic insulin release and reduces the insulin sensitivity of muscle. SGLT2-specific antibodies recognize a protein in muscle of similar size to the kidney SGLT2; this protein localizes to the muscle t-tubules, together with Na,K-ATPase-2 and MAP17, the regulatory subunit of SGLT2. However, skeletal muscles do not express a full-length transcript of Slc5a2 (SGLT2), and SGLT2-specific inhibitors do not inhibit mSGLT with high affinity. The novel transporter may be a muscle variant of Slc5a2 that results from post-transcriptional or post-translational mechanisms. mSGLT and its regulation offer potential muscle-specific therapeutic targets for treating hyperglycemia and other conditions when insulin-stimulated glucose disposal into muscle is impaired.