FAF1 and FAF2 enhance human p97-UFD1-NPL4 complex unfoldase activity enabling rational design of p97 activators
FAF1 and FAF2 enhance human p97-UFD1-NPL4 complex unfoldase activity enabling rational design of p97 activators
Dasgupta, P.; Kelsall, I. R.; Anand, G.; Perez-Rafols, A.; Knebel, A.; Gourlay, R.; Masson, G. R.; Kulathu, Y.
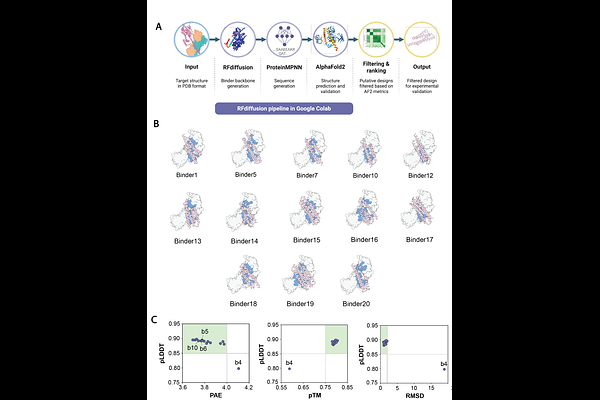
AbstractVCP/p97 is an AAA+ ATPase that, together with its cofactors UFD1-NPL4 (p97-UN), binds and unfolds ubiquitylated substrates to maintain cellular homeostasis. The human p97-UN complex associates with additional cofactors, but how these cofactors modulate p97-UN activity is not fully understood. Here, we screen for cofactors that enhance p97-UN activity and identify FAF2 as the strongest activator. Using biochemical and structural approaches, we show how FAF2 engages p97-UN and polyubiquitin to promote unfolding. We define a conserved activation motif in FAF2 that contacts both UFD1 and the ubiquitin proximal to the initiator, stabilizing and initiating unfolding in a UFD1-dependent manner. We leverage the features of FAF2 AM to engineer de novo proteins that potently enhance unfolding, providing a rational strategy to boost p97 activity. Our findings reveal how cofactors can provide additional adaptive control, fine-tuning human p97 activity to unfold challenging substrates and those modified with short ubiquitin chains.