HIF-1α coordinates adrenal steroidogenesis through direct transcriptional control and regulation of miRNA biogenesis
HIF-1α coordinates adrenal steroidogenesis through direct transcriptional control and regulation of miRNA biogenesis
Stepien, B. K.; Sinha, A.; Ariyeloye, S.; Krueger, A.; Mirtschink, P.; Bartoszewski, R.; Wielockx, B.
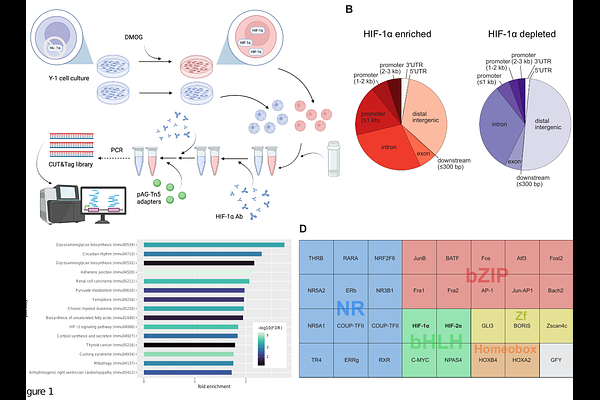
AbstractBackground Adrenal steroid hormone production is essential for systemic stress adaptation and metabolic homeostasis, and it is tightly regulated by oxygen availability. Previously, we demonstrated that acute hypoxia suppresses adrenal steroidogenesis through HIF-1-dependent induction of microRNAs (miRNAs) that target key steroidogenic enzymes. However, the mechanisms by which HIF-1 controls miRNA expression and activity in this context remain unclear. Methods To address this issue, we mapped the genome-wide HIF-1 binding landscape in murine adrenocortical cells using Cleavage Under Targets & Tagmentation (CUT&Tag). We integrated this data with gene expression analyses following pharmacological HIF-1 stabilization, physiological hypoxia, and genetic HIF-1 depletion to distinguish HIF-1-dependent effects from broader hypoxia-driven responses. Results We detected HIF-1 binding at loci encoding steroidogenic enzymes and steroidogenesis-associated miRNAs. Unexpectedly, we also detected binding at genes involved in miRNA biogenesis and function, including components of the nuclear microprocessor complex and the cytoplasmic RNA-induced silencing complex (RISC). Functional analyses revealed that hypoxia broadly represses the expression of miRNA-processing genes through both HIF-1-dependent and -independent mechanisms. Notably, HIF-1 selectively modulated or counteracted this repression in a gene-specific manner, indicating a regulatory role beyond direct transcriptional activation. Conclusions These findings reveal an unrecognized layer of hypoxia-driven cell communication, wherein HIF-1 coordinates the transcriptional and post-transcriptional regulation of adrenal steroidogenesis by shaping the miRNA-processing landscape. This work extends our understanding of how oxygen-sensitive signaling pathways integrate gene expression and RNA-based regulatory mechanisms to control endocrine function.