Mechanisms of MCM2-7 helicase activation and initial DNA melting at near base-pair resolution
Mechanisms of MCM2-7 helicase activation and initial DNA melting at near base-pair resolution
Weekes, C.; Willerding, L.; Khadayate, S.; Liebl, K. B. M.; Mossler, A.; Montoya, A.; Rauthe, V.; Karimi, M. M.; Zacharias, M.; Ulrich, H. D.; Speck, C.; Reuter, L. M.
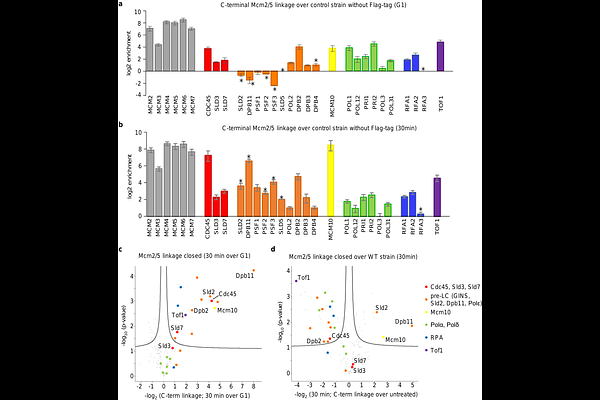
AbstractDuring eukaryotic DNA replication initiation, inactive MCM2-7 double-hexamers assembled at replication origins must be converted into two active CMG helicases, yet how this transition is coupled to origin DNA unwinding in vivo remains unclear. Here, we identify a DNA-bound intermediate with an extended genomic footprint that forms during helicase activation. Genome-wide mapping of initial strand separation reveals that DNA unwinding initiates near the N-terminal interface of opposing MCM2-7 hexamers. At these sites, the origin DNA exhibits a conserved AT-rich/GC-rich/AT-rich sequence architecture on which the helicase complex is centred, consistent with a role in promoting DNA extrusion following double-hexamer separation. We further show that restricting hexamer rotation and splitting delays release of the Cdc45-loading factor Sld3, demonstrating that mechanical transitions during helicase activation are tightly coupled to complex disassembly. Finally, we provide in vivo evidence that single-stranded DNA is ejected through a specialised DNA exit gate at the Mcm2/5 interface during helicase activation, which is dispensable for ongoing DNA synthesis. Together, these findings establish a mechanistic framework for how replication origins are remodelled to initiate DNA replication and reveal key intermediates and DNA transactions during helicase activation.