Active microbial communities and their extrachromosomal elements link organic matter degradation to methane cycling in anoxic sediments
Active microbial communities and their extrachromosomal elements link organic matter degradation to methane cycling in anoxic sediments
Dede, B.; Zehnle, H.; Skoog, E.; Priest, T.; Beck, K.; Buergmann, H.; Schoelmerich, M. C.
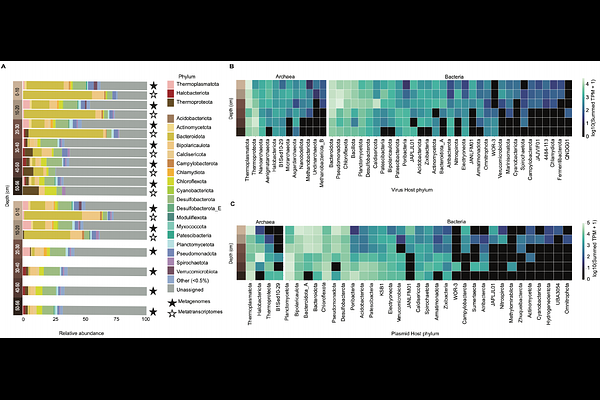
AbstractAnaerobic carbon transformation in freshwater sediments drives substantial methane emissions globally, yet the microbial taxa linking complex carbon degradation to methane production remain poorly characterized. Here, we combined metagenomics with the first metatranscriptomic dataset from the anoxic sediments of meromictic Lake Cadagno (Swiss Alps) to identify the active microbial clades, metabolic pathways, and extrachromosomal elements (ecDNA) across a depth gradient within the upper 56 cm of sediment. We recovered 802 species-level metagenome-assembled genomes (MAGs) spanning 66 phyla and identified a Bacteroidota clade (VadinHA17) as one of the most abundant and transcriptionally active populations in the sediment. This clade encodes and transcribes a broad range of diverse glycoside hydrolases (GH), indicating a central role in complex carbohydrate degradation. Transcriptional profiles suggest that this clade ferments organic substrates to acetate and hydrogen, which are key substrates for methanogenesis. In line with this, the acetoclastic methanogen Methanothrix and hydrogenotrophic Methanoregula were among the most abundant and transcriptionally active archaea in the same depth layers. Beyond microbial genomes, we detected 86,905 viral OTUs (vOTUs) and 2,136 plasmid OTUs (pOTUs), with free viruses and plasmids accounting for 5-10% and 0.2% of all sequencing reads, respectively. Notably, plasmids and viruses associated with Bacteroidota VadinHA17 encode and transcribe GHs that could augment host carbohydrate-degrading capacity. Together, these findings reveal new details on how methane production in anoxic lake sediments emerges from a network spanning primary fermentation, methanogenesis and ecDNA-mediated metabolisms.