Interplay of the ribosome A and CAR sites
Interplay of the ribosome A and CAR sites
Raval, M.; Zhou, Y.; Lynch, M.; Krizanc, D.; Thayer, K.; Weir, M. P.
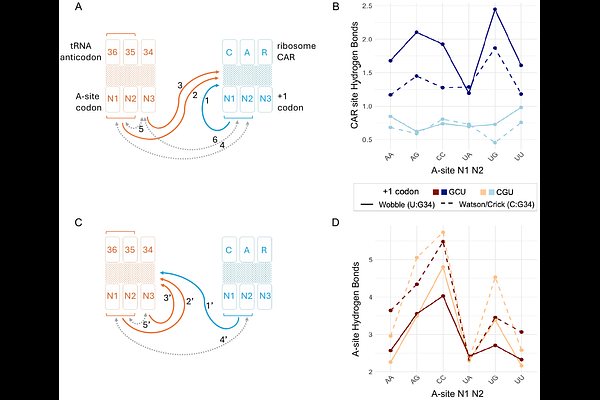
AbstractProtein translation is a highly regulated process influenced by multiple factors at the initiation, elongation, and termination stages. One notable regulatory element of the ribosome is the CAR interaction surface, a three-residue motif in the structure of the ribosome composed of C1274 and A1427 of S. cerevisiae 18S rRNA (corresponding to C1054 and A1196 in E. coli 16S rRNA) and R146 of ribosomal protein Rps3. CAR is highly conserved and positioned adjacent to the amino-acyl (A site) decoding center. It establishes hydrogen bonds with the +1 codon next in line to enter the ribosome A site, acting as an extension of the tRNA anticodon and forming base-stacking interactions with nucleotide 34 of the tRNA. However, despite CAR's enzymatically strategic positioning within the ribosome, its functional relationship with the A site remains poorly characterized. Using molecular dynamics (MD) simulations, we examined the interplay between the A site and CAR site, revealing sequence-dependent modulation of H-bonding and pi-stacking interactions within and between the two sites. These findings highlight the interplay between the A site and CAR site, suggesting a structural and functional connection between these two regions of the ribosome that may contribute to mRNA sequence-specific tuning of translation elongation.