Mining the heparinome for cryptic antimicrobial peptides that selectively kill gram-negative bacteria
Mining the heparinome for cryptic antimicrobial peptides that selectively kill gram-negative bacteria
Sandin Garcia, D.; Valle, J.; Gomez, J.; Comas, L.; Larrosa, M. N.; Gonzalez, J. J.; Jimenez, M. A.; Andreu, D.; Torrent, M.
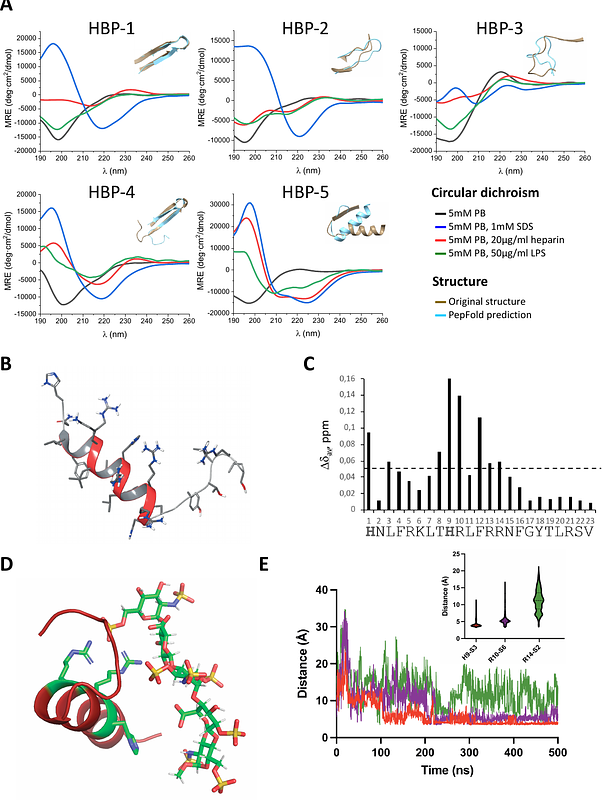
AbstractGlycosaminoglycan (GAG)-binding proteins regulating essential processes such as cell growth and migration are essential for cell homeostasis. As both GAGs and the lipid A disaccharide core of gram-negative bacteria contain negatively charged disaccharide units, we hypothesized that GAG-binding proteins could also recognize LPS and enclose cryptic antibiotic motifs. Here, we report novel antimicrobial peptides (AMPs) derived from heparin-binding proteins (HBPs), with specific activity against gram-negative bacteria and high LPS binding. We used computational tools to locate antimicrobial regions in 82% of HBPs, most of those colocalizing with putative heparin binding sites. To validate these results, we synthesized five candidates [HBP1-5] that showed remarkable activity against gram-negative bacteria, as well as a strong correlation between heparin and LPS binding. Structural characterization of these AMPs shows that heparin or LPS recognition promotes a conformational arrangement that favors binding. Among all analogs, HBP-5 displayed the highest affinity for both heparin and LPS, with antimicrobial activities against gram-negative bacteria at the nanomolar range. These results suggest that GAG-binding proteins are involved in LPS recognition, which allows them to act also as antimicrobial proteins. Some of the peptides reported here, particularly HBP-5, constitute a new class of AMPs with specific activity against gram-negative bacteria.