Convergence on BRAF and MAPK Signaling in Glioma Development in a P53-ENU model
Convergence on BRAF and MAPK Signaling in Glioma Development in a P53-ENU model
Desai, K.; Tymofyeyeva, A.; Javier, M.; Svergun, N.; Ye, V.; Bullivant, G.; Forst, J.; Ware, M.; Kushida, M.; Lee, L.; Yu, C.; Whetstone, H.; Ward, R.; Dave, B. M.; Aman, A.; Dirks, P. B.
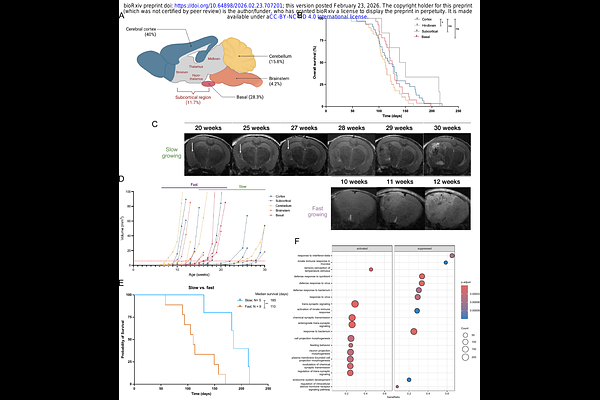
AbstractPediatric high-grade gliomas (HGGs) are aggressive and lethal brain tumors that account for 15-20% of all pediatric central nervous system (CNS) malignancies and remain largely incurable. These tumors, despite having mutational targets that activate MAPK signaling, are frequently resistant to targeted therapies in their malignant states, but often show responses when they are in lower grade form. These findings suggest a need to identify and intercept early tumorigenic events that arise in earlier tumor developmental stages. To investigate the molecular events in glioma progression, we developed and characterized a NestinCre/+;Trp53fl/fl mouse model combined with in utero exposure to the classic chemical mutagen N-ethyl-N-nitrosourea (ENU). This model mimics the context of genetic predisposition paired with an environmental genotoxic insult, and mice with loss of TRP53 in early neural precursors who are exposed to ENU at embryonic day 13.5 reproducibly develop HGGs postnatally that retain features of the human tumors. By sampling discrete lesions at premalignant, early, and late stages, we observed progressive increases in genetic complexity, stemness features, and immune signatures across tumor evolution. Notably, a recurrent Braf mutation emerged in a majority of early lesions and persisted in advanced tumors, consistent with the occurrence of BRAFV600E mutations in human gliomas that arise in children and undergo transition from lower grade to higher grade stages. Additional components of the RAS-RAF-MAPK signaling cascade, including Kras, Nras and Nf1 were found to be mutated at the late tumor stage, indicating convergent activation of this pathway in this model. Cell lines derived from early lesions responded to BRAF inhibitors, but cells from endpoint tumors were less responsive. Together, this model reveals aspects of the molecular and cellular evolution of glioma development in vivo, and identifies MAPK signalling as a critical molecular bottleneck selected for the ENU-induced mutations. This genetic-environmental model may be valuable to understanding key determinants of glioma initiation and progression, and for evaluating new therapies that limit MAPK signaling.