Sub-cellular Systems Drift Drives Mosaic Evolution of Mammalian Neurons.
Sub-cellular Systems Drift Drives Mosaic Evolution of Mammalian Neurons.
Rosario, J. G.; Kim, J.
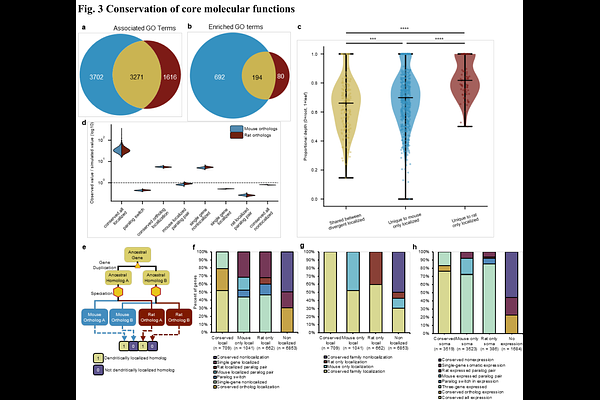
AbstractEvolution of the mammalian brain has been described as mosaic evolution wherein natural selection for behavioral function promotes independent evolution of specific functional units despite developmental constraints that might govern overall change(Barton & Harvey, 2000; DeCasien & Higham, 2019). Evidence of mosaic evolution has been reported at the level of gene expression in individual structures(Kautt et al., 2025), cell type abundances(Chen et al., 2025), as well as gene regulatory changes at the single cell level(Caglayan et al., 2023; Hao et al., 2024; Suresh et al., 2023). In particular, it has been hypothesized that brain evolution involves changes in circuit organization(Suresh et al., 2023; Thiebaut de Schotten & Forkel, 2022). Circuit-level changes involve sub-cellular compartments that mediate synaptic activity, raising the question whether mosaic brain evolution might be found at the sub-cellular scale. Here, we examine the rate of evolutionary divergence between Mus musculus (C57BL/6) and Rattus norvegicus (Sprague-Dawley) for their dendritic transcriptome, which shapes the post-synaptic proteome through sub-cellular localization and local translation(Bourke et al., 2023). We address the problem of variable assessment of the dendritic transcriptome by micro-dissecting individual hippocampal pyramidal neurons to create matched single cell libraries of the soma and the dendrites from the same cell and apply a machine learning model to predict localization. Our results show that the dendritic transcriptome is significantly more divergent than the soma, but the core functional roles of the dendritically localized genes are conserved. Examining gene families for their localization suggests enrichment of family level conservation or localization. We propose that the observed divergence may arise from a combination of adaptive modulation and system drift under selection for core function. Our study suggests fine-grained mosaic evolutionary dynamics at the scale of synaptic function that mediates information processing and neural connectivity.