Model-Driven Hybrid AI Framework for End-to-End Autonomous Decision-Making in Drug Development
Model-Driven Hybrid AI Framework for End-to-End Autonomous Decision-Making in Drug Development
Lee, H.; Kang, H.; Jung, W.; Cho, H.; Goo, S.; Yun, H.-y.; Kim, M.-G.; CHAE, J.-w.; Park, S.-M.; Lee, S.; Kim, J. H.; Jung, S.
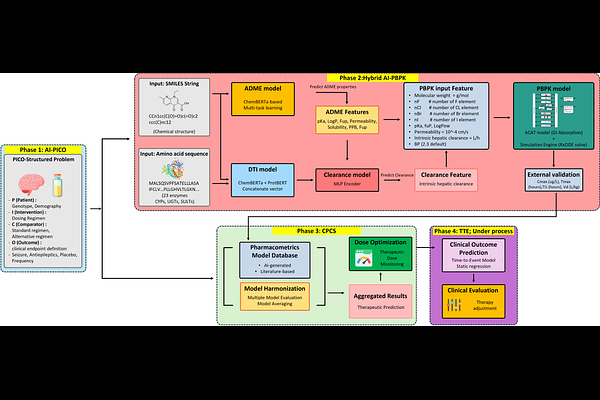
AbstractDecision-making in drug development spans heterogeneous stages from molecular design to clinical optimization, yet computer-aided workflows across stages remain fragmented, limiting traceability from evidence-derived clinical questions to simulation scenarios and decision endpoints. We present a model-driven hybrid AI framework for end-to-end decision support that treats PICO (Participants, Intervention, Comparison, Outcome) as a machine-actionable specification to define scenarios and align endpoints across modules. Given a PICO-defined clinical question and a drug SMILES, the framework infers PBPK-ready parameters from structure, generates exposure via mechanistic PBPK simulation, and links exposure outputs to clinic-facing therapeutic drug monitoring (TDM) decision support using an open-source NLME-based Clinical Pharmacokinetic Consultant Service (CPCS). We additionally implement an LLM-based PICO extraction layer to structure clinical abstracts into P/I/C/O elements for systematic evidence ingestion. Results include backbone verification for DTI-guided intrinsic clearance prediction under fold-error bounds and PBPK trajectory plausibility, and external validation of the hybrid AI-PBPK pipeline showing substantial dispersion for several endpoints. An input-controlled comparison between PhysioSim and PK-Sim on N = 9 drugs yields similarly degraded accuracy under identical AI-predicted inputs, suggesting upstream parameter quality as a dominant constraint. For evidence ingestion, prompting-only PICO extraction achieves F1 of 0.687 on EBM-NLP and 0.552 on TB-PICO. For clinical utility, CPCS improves TDM performance over a PKS baseline on phenobarbital and vancomycin, reducing MAPE by approximately 26-60% depending on configuration. Overall, the framework provides a modular, traceable blueprint linking evidence-defined questions to exposure simulation and TDM decision support while preserving mechanistic interpretability. Future work will integrate outcome-aligned time-to-event modeling and federated learning to close the PICO-to-outcome loop and improve parameter inference under multi-site governance.