mSWI/SNF complex inhibition sensitizes KRAS-mutant lung cancers to targeted therapies via epithelial-mesenchymal subversion
mSWI/SNF complex inhibition sensitizes KRAS-mutant lung cancers to targeted therapies via epithelial-mesenchymal subversion
Gentile, C.; Feng, W. W.; Lenahan, S. M.; Ying, A. W.; Card, D. C.; Wu, F. T. H.; Pham, N.-A.; Radulovich, N.; Cao, P. M.; Hueniken, K.; Li, Q.; Tsao, M.-S.; Kulesza, J.; Hinkley, M. M.; Liao, L.; Tsai, J. A. < A.; Kohler, J.; Facchinetti, F.; Li, J.; Weston, C.; Locquet, M.-A.; Ngo, K.; Gokhale, P. C.; Sacher, A. G.; Janne, P. A.; Kadoch, C.
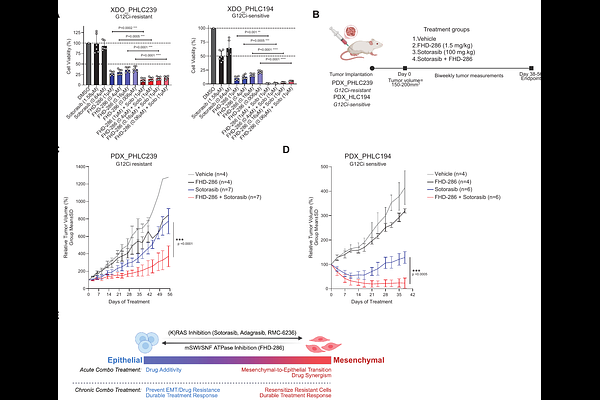
AbstractTargeted therapies for KRAS-mutant non-small lung cancer (NSCLC) have shown promising clinical results, however, incomplete tumoral responses and the inevitable emergence of therapeutic resistance remain critical challenges. Here we identify mSWI/SNF chromatin remodeling complexes as critical determinants of (EMT)-mediated KRAS inhibitor inefficacy and resistance in KRAS G12C lung cancers. Treatment with the clinical-grade SMARCA4/2 inhibitor, FHD-286, dampens EMT-mediated acquired resistance in drug-responsive models and similarly resensitizes drug-refractory models by rewiring mSWI/SNF chromatin localization and activities that modulate epithelial transcriptional programs and cell state. Further, synergistic mSWI/SNF and KRAS inhibitor combination treatment sensitizes non-G12C KRAS-mutant NSCLC cells to pan-RAS and G12D-specific inhibitors. Finally, FHD-286 and sotorasib combination treatment results in potent anti-tumor efficacy in both G12Ci-resistant and -sensitive organoid models and in vivo patient-derived xenograft (PDX) systems. These data nominate mSWI/SNF inhibition as a combination strategy to improve KRAS inhibitor efficacy, response duration, and to mitigate emergence of resistance.