PROTAC internalization and target degradation require clathrin-mediatedendocytosis
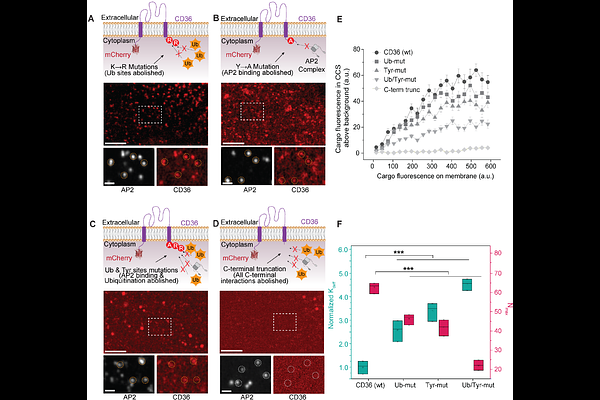
PROTAC internalization and target degradation require clathrin-mediatedendocytosis
Liu, H.-Y.; Wang, Z.; Sharma, R.; Perez, J.; Kusaj, B.; Zhou, H.; Wang, M.; Huibregtse, J.; Li, H.-Y.; Stachowiak, J.
AbstractProteolysis-targeting chimeras (PROTACs) are emerging as potent tools for targeted protein degradation that overcome many of the limitations of traditional small molecule inhibitors. Yet how these hetero-bifunctional therapeutics enter cells remains a mystery. While passive diffusion is conventionally assumed, the bulky structure of PROTACs suggests that active transport may be required. Recently, the fatty acid transporter CD36 was identified as a key receptor for PROTACs. However, because the uptake mechanism of CD36 is itself unknown, how PROTACs enter cells remains a mystery. Here we show that PROTAC uptake and function require clathrin-mediated endocytosis. We uncover previously unrecognized clathrin adaptor binding motifs in the CD36 C-terminus and use live-cell imaging to visualize the recruitment of both CD36 and PROTACs to sites of clathrin-mediated endocytosis on the cellular plasma membrane. Strikingly, disruption of clathrin assembly through either genetic or pharmacological means abolishes all detectable PROTAC-induced protein degradation, demonstrating that the clathrin pathway is required for the function of PROTACs that utilize diverse E3 enzymes against multiple targets. These results elucidate the molecular mechanism of PROTAC entry into cells, providing critical information for optimizing cellular uptake and response to targeted degraders.