Topological Environment in Genetic and Metabolic Networks
Topological Environment in Genetic and Metabolic Networks
Castillo-Villalba, M. P.
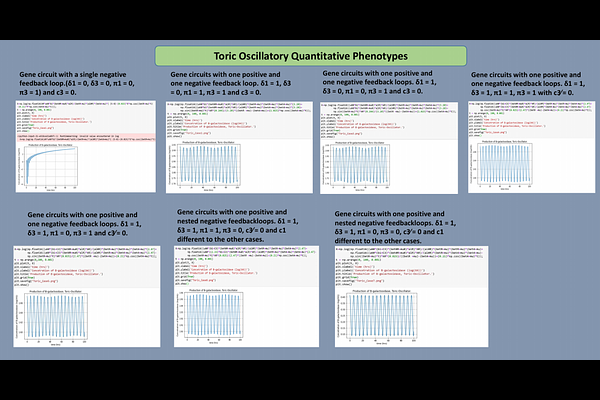
AbstractThe analysis of large gene and metabolic networks is often hindered by unknown biochemical parameters and the nonlinear nature of classical S-system models. To address this, we introduce a framework based on combinatorial toric geometry computed with tools such as Normaliz, SageMath, it is worth mentioning this technique in not restrictive to integer vectors, there exists a natural extension to real geometries. Unlike traditional approaches, which rely on parameter dependent fixed points, our method constructs a Topological Environment derived from the dual space of kinetic orders, leading to what we call orthogonal enzyme kinetics. Within this topological setting, fixed points are computed on the algebraic torus, enabling the transformation of nonlinear dynamics into linear forms. Importantly, these fixed points are independent of kinetic parameters and depend only on network topology and interaction signs. Applying this methodology to gene circuits involved in circadian rhythms, we reproduce previously reported oscillatory physiologies.