Discovery of the first small-molecule extracellular inhibitor of KCa3.1
Discovery of the first small-molecule extracellular inhibitor of KCa3.1
Massa, J.; Hense, J.; Gangnus, T.; Gozzi, M.; Bulk, E. E.; Burckhardt, B.; Duefer, M.; Schwab, A.; Koch, O.
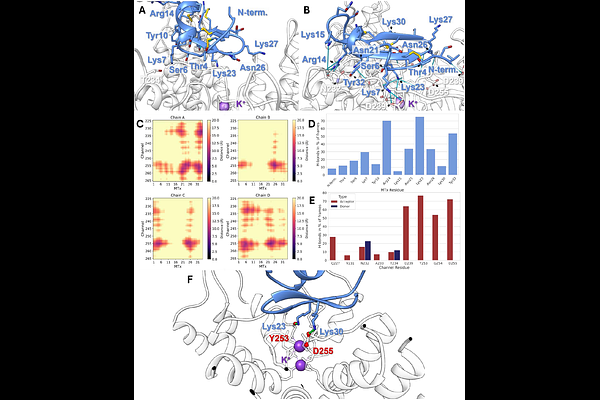
AbstractThe ion channel KCa3.1 plays a role in immune regulation, red blood cell function, and is linked to numerous types of cancer. Various animal toxins, such as maurotoxin, bind to the extracellular side of KCa3.1, providing a potential starting point for inhibitor development. We report in this work the discovery of a novel, small-molecule inhibitor, with a micromolar IC50, which was specifically designed to target plasma-membrane KCa3.1 channels from the extracellular side. This compound can serve as a starting point for the development of more selective inhibitors and probes. For the identification of new extracellular inhibitors, molecular dynamics simulations were performed using the experimental structures of KCa3.1 and maurotoxin. The simulations produced a validated binding mode, highlighting key residues involved in the interaction between the toxin and the channel. These findings laid the foundation for the structure-based identification of novel extracellular small-molecule inhibitors of KCa3.1. The Molport database, containing approximately 50 million compounds, was screened using protein-ligand docking, yielding a hit molecule that was experimentally confirmed using patch clamp assays.