Beneficial effects of intermittent fasting in NASH and subsequent HCC development are executed by concerted PPAR alpha and PCK1 action in hepatocytes
Beneficial effects of intermittent fasting in NASH and subsequent HCC development are executed by concerted PPAR alpha and PCK1 action in hepatocytes
Gallage, S.; Ali, A.; Barragan Avila, J. E.; Seymen, N.; Ramadori, P.; Joerke, V.; Zizmare, L.; Kosla, J.; Li, X.; Focaccia, E.; Yousuf, S.; Sijmonsma, T.; Rahbari, M.; Kommoss, K. S.; Billeter, A.; Prokosch, S.; Rothermel, U.; Mueller, F.; Hetzer, J.; Heide, D.; Machauer, T.; Malek, N. P.; Longerich, T.; Rose, A. J.; Roth, S.; Schwenck, J.; Trautwein, C.; Karimi, M. M.; Heikenwalder, M.
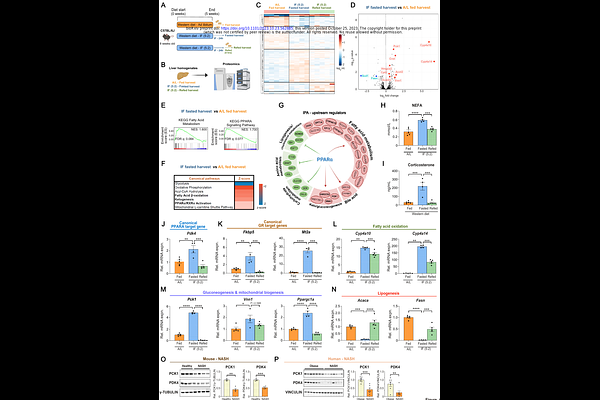
AbstractThe role and molecular mechanisms of intermittent fasting (IF) in non-alcoholic steatohepatitis (NASH) and its transition to hepatocellular carcinoma (HCC) are unknown. Here, we identified that an IF 5:2 regimen (two non-consecutive days of food deprivation per week), initiated in the active phase of mice, prevents/ameliorates NASH and fibrosis as well as reduces subsequent HCC development without affecting total calorie intake. The timing, length and number of fasting cycles as well as the type of NASH diet were all critical parameters determining the effectiveness of the fasting benefits. Combined proteomic, transcriptomic and metabolomic analyses identified that PPAR alpha and glucocorticoid receptor (GR)-PCK1 act co-operatively as hepatic executors of the fasting response by promoting fatty acid catabolism and gluconeogenesis whilst suppressing anabolic lipogenesis. In line, PPAR alpha targets and PCK1 were reduced in human NASH. Additionally, dynamic [18F]FDG-PET analysis in vivo revealed increased [18F]FDG uptake/retention and enhanced gluconeogenesis in the liver upon fasting (in accordance with PPAR alpha and GR-PCK1 activation) when assessed by compartmental modelling. Hepatocyte-specific GR deletion only partially abrogated the hepatic fasting response. In contrast, the combined knockdown of Ppara and Pck1 in vivo abolished the beneficial outcomes of fasting against inflammation and fibrosis, confirming their causal relationship in integrating systemic signalling in hepatocytes. Notably, PPAR alpha agonist pemafibrate recapitulated key aspects of hepatic fasting signalling at a molecular level. Therefore, IF or pharmacological mimetics of the PPAR alpha and/or GR-PCK1 axis could be a viable intervention against NASH and subsequent liver cancer.