Local Tumor Microenvironment Niches Correlate With Survival And Immunotherapy Response In Human Glioblastoma
Local Tumor Microenvironment Niches Correlate With Survival And Immunotherapy Response In Human Glioblastoma
Petitprez, F.; Webb, S.; Morrison, G.; Merotto, L.; Webb, J.; Xie, Y.; Guney, E.; Weiss, W. A.; Finotello, F.; Kitamura, T.; Pollard, S. M.; Pollard, J. W.
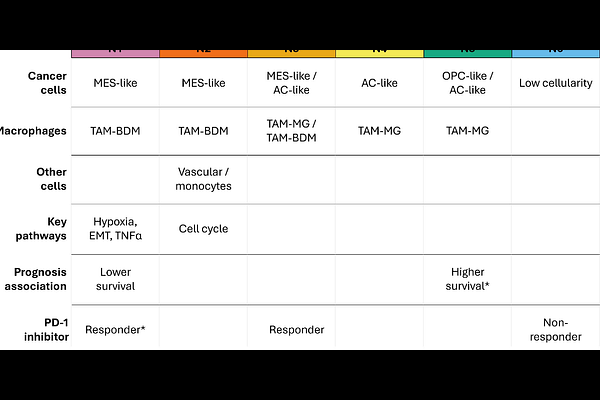
AbstractBackground: Glioblastoma (GBM) is an aggressive form of primary brain cancer. Recent efforts to characterize GBM using single-cell or spatially-resolved transcriptomics have revealed a tremendous intra-tumoral heterogeneity between malignant cells and between different tumor areas. However, most efforts have focused on malignant cells, and the spatial and cellular heterogeneity of the tumor microenvironment (TME) remains poorly understood. Moreover, it is unclear how TME compositions and organizations influence clinical outcomes for patients. Results: Integrating spatial transcriptomics, single-cell RNA-seq and histology on 25 tumors, cellular composition of the TME was estimated on over 46,000 55-m wide spots. Spatial associations were revealed between mesenchymal-like cancer cells and monocyte-derived macrophages. Spots were clustered into six unique classes of TME, exhibiting differential composition of malignant and immune cells, and distinct activation of biological pathways. Spatial transcriptomics-informed deconvolution of large-scale bulk RNA-seq datasets revealed that the niche composition of tumors associated significantly with patient survival and response to immunotherapy. Mesenchymal-like, monocyte-derived macrophages-rich and hypoxic niche N1 associated with lower overall survival while oligodendrogial progenitor-like and microglia-derived macrophages-enriched niche N5 is associated with longer patients survival. Analysis of data from patients treated with immunotherapy showed that niches N1 and mixed mesenchymal-like and astrocyte-like niche N3 associated with response to PD-1 inhibitors. Conclusions: Our results show that GBM exhibits a strong spatial heterogeneity of TMEs, with distinct categories of niche. The niche composition of tumors associated with survival and immunotherapy response. Our results suggest incorporation of TME niches as biomarkers for risk stratification and therapeutic decisions for patients.