Novel Plasma Proteomics and Phosphoproteomics Platform Captures Pleiotropic Cardiometabolic Spectrum Effects of Semaglutide in Patients with T2D and Atherosclerosis: A Companion Diagnostic Pilot Study from the STOP (Semaglutide Treatment On coronary atherosclerosis Progression) Randomized Trial
Novel Plasma Proteomics and Phosphoproteomics Platform Captures Pleiotropic Cardiometabolic Spectrum Effects of Semaglutide in Patients with T2D and Atherosclerosis: A Companion Diagnostic Pilot Study from the STOP (Semaglutide Treatment On coronary atherosclerosis Progression) Randomized Trial
Manousopoulou, A.; White, C. H.; Hamal, S.; Nihalani, R.; Budoff, M. J.; Garbis, S. D.
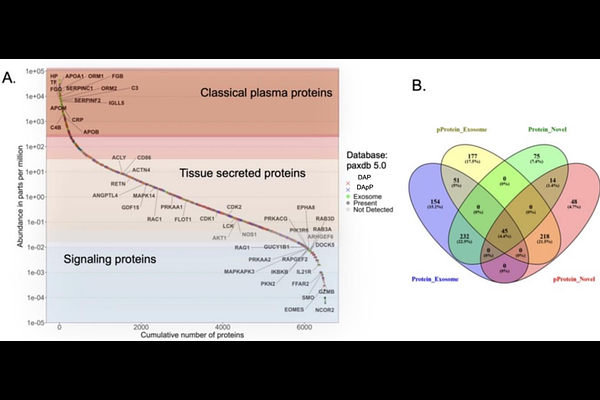
AbstractBACKGROUND: As a GLP1 R agonist, semaglutide is known to exhibits pleiotropic health effects across the cardiometabolic spectrum in patients with type 2 Diabetes Mellitus (T2D). However, in depth and unbiased protein and phosphoprotein level evidence that reflects such effects of semaglutide in plasma remains elusive. OBJECTIVES: This pilot study applied an innovative plasma proteomics and phosphoproteomics technology to a sub-set of patients with T2D that participated in the Semaglutide Treatment effect on coronary atherosclerosis Progression (STOP) randomized trial. The aim of this study was to identify the systemic effects of semaglutide treatment in pathways that underpin its pleiotropic cardiometabolic spectrum health benefits. METHODS: The study applied a proprietary liquid biopsy discovery proteomics platform and its derivative cardiometabolic spectrum database (International patent PCT/US2021/063407) to 16 patients from the STOP randomized trial. Plasma samples from 8 patients in the active group and 8 patients in the placebo group at baseline and 52 weeks post treatment were analyzed. The methodology entailed the use of a unique liquid fixative chemistry to instantly solubilize and stabilize plasma proteins and phosphoproteins at room temperature followed by their direct microflow monolithic partition chromatography, dialysis purification, solution phase proteolysis, multiplex isobaric stable isotope labeling of proteotypic peptides, lab-on-chip TiO2/ZrO2 phosphopeptide enrichment and nanotechnology enhanced ultra-high resolution LC-MS analysis. To identify differentially abundant proteins (DAPs) and phosphoproteins (DApPs) in patients treated with semaglutide vs. placebo, the respective abundance ratio for each was considered repectively. Ratios were log2-transformed to normalize their distribution. A one-sample T-Test (paired) using the two-stage Benjamini Yekutieli Krieger step up method for multiple hypothesis testing FDR correction of the p-value was performed. The threshold of significance was set at q<0.05. DAPs/DApPs were corrected for placebo. The expressed proteome and phosphoproteome were further interpreted with a multifactorial computational biology pipeline to deconvolute their underlying protein-level molecular pathways and their networks along with transcriptional factors and kinases that regulate them. RESULTS: This study achieved an extremely high depth-and-breadth in quantitative proteome and phosphoproteome coverage from only 20uL whole plasma equivalent from each patient. Namely, a total of 13,173 proteins and 25,578 phosphopeptides were fully profiled (q<0.05). Of these, 1,040 were differentially abundant proteins (DAPs) and 1,064 were differentially abundant phosphoproteins (DApPs), in the semaglutide treated group after correcting for placebo, at an absolute log2-fold-change of >0.5, CV<15%, q< 0.05. Of interest, this study profiled over 85% of all proteins/phosphoproteins (6,700) reported to date based on the use of the well curated and up-to-date PaxDB database. Over 70% of these known proteins were of exosomal origin. Importantly, an additional ~9000 plasma proteins and phosphoproteins of this study constituted entirely novel observations. Contextualization of the DAPS, DApPs, kinases and transcription factors for all statistically significant enriched canonical pathways (FDR-corrected q<0.001) revealed a wide array of pathophysiological processes attributed to semaglutide treatment. Furthermore, these pathways provided a molecular understanding to the reported imaging biomarkers against the same STOP trial patients. CONCLUSIONS: This feasibility study demonstrated how an effective plasma proteomics and phosphoproteomics platform can generate a treatment-adaptive companion diagnostic molecular signature that holistically captures the multiple cardiometabolic spectrum health effects of semaglutide in patients with atherosclerosis and T2D.