Biosensor-Driven Strain Engineering Reveals Key Cellular Processes for Maximizing Isoprenol Production in Pseudomonas putida
Biosensor-Driven Strain Engineering Reveals Key Cellular Processes for Maximizing Isoprenol Production in Pseudomonas putida
Menasalvas, J.; Kulakowski, S.; Chen, Y.; Gin, J. W.; Turumtay, E. A.; Apolonio, M. A.; Baral, N. R.; Rivier, A.; Yunus, I. S.; Garber, M. E.; Scown, C. D.; Adams, P. D.; Lee, T. S.; Blaby, I. K.; Baidoo, E. E. K.; Petzold, C. J.; Eng, T.; Mukhopadhyay, A.
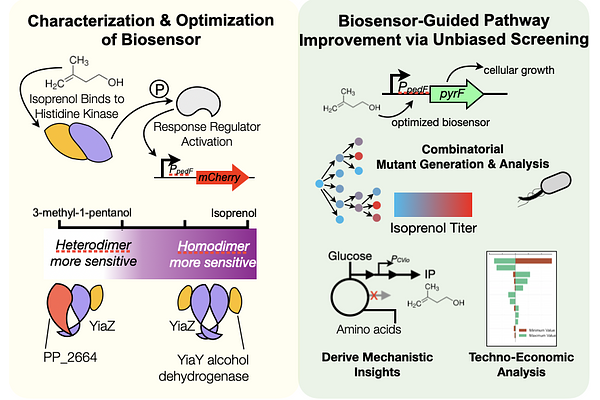
AbstractSynthetic biology tools have accelerated the generation of simple mutants, but combinatorial testing remains a major hurdle. High-throughput methods struggle translating from proof-of-principle molecules to advanced bioproducts. We address this challenge with a biosensor-driven strategy for enhanced isoprenol production in Pseudomonas putida, a key precursor for sustainable aviation fuel and platform chemicals. This biosensor leverages P. putida\'s native response to short-chain alcohols via a previously uncharacterized hybrid histidine kinase signaling cascade. Refactoring the biosensor for a conditional growth-based selection enabled identification of competing cellular processes with a ~16,500-member CRISPRi-library. An iterative combinatorial strain engineering approach yielded an integrated P. putida strain producing ~900 mg/L isoprenol in glucose minimal medium, a 36-fold increase. Ensemble -omics analysis revealed metabolic rewiring, including amino acid accumulation as key drivers of enhanced production. Techno-economic analysis elucidated the path to economic viability and confirmed the benefits of adding amino acids outweigh the additional costs. This study establishes a robust biosensor driven approach for optimizing other heterologous pathways, accelerating microbial cell factory development.