From the lung to the muscle: Systemic insights from an integrative MultiOmics analysis of harbour porpoises in poor respiratory health
From the lung to the muscle: Systemic insights from an integrative MultiOmics analysis of harbour porpoises in poor respiratory health
Dönmez, E. M.; Siebels, B.; Drotleff, B.; Nissen, P.; Derous, D.; Fabrizius, A.; Siebert, U.
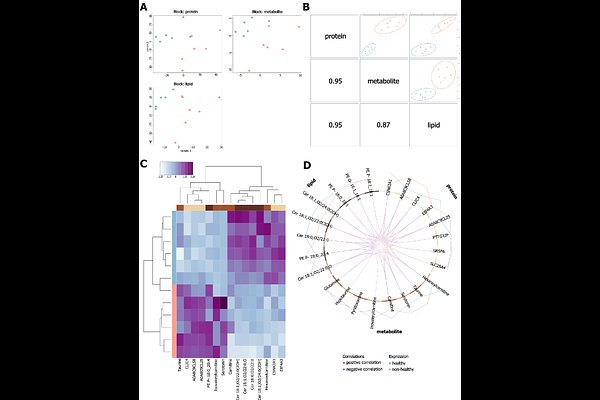
AbstractHarbour porpoises (Phocoena phocoena) in the North and Baltic Seas are increasingly impacted by anthropogenic pressures, including underwater noise, fisheries and pollution. These pressures correlate with declining population health, particularly affecting the respiratory system. Growing pathological lesions, partly resulting from high prevalence of parasitic infestations and subsequent diseases, can impair tissue function and oxygen supply to distant end-organs. In this study, we applied an integrative MultiOmics approach (proteomics, metabolomics, lipidomics) to analyse the lungs and muscles of 12 wild harbour porpoises with compromised respiratory health. Our aim was to identify dysregulated biological pathways across omics layers to advance insights into adaptive physiological responses and to define disease-associated molecular signatures that could assist health assessments. Our analysis revealed pronounced immune system and antioxidative responses in the lungs and muscles, indicated by enhanced immunoglobulins, plasmalogens and glutathione-related proteins. In the lungs, high cardiolipin levels and reduced collagen suggest impaired tissue structure and function, while tissue maintenance processes were elevated in the muscle. Both tissues exhibited metabolic alterations suggestive of energetic imbalance, including increased purine metabolism in the lung and decreased lipid metabolism in the muscle. Several dysregulated molecules were shared across tissues, pointing to pathophysiological effects. The proposed disease-associated molecular signatures included the protein SLC25A4, the metabolite O-phosphoethanolamine and the lipid TG O-16:0_16:0_20:4 for the lung, and the protein SPEG, the metabolite pipecolic acid, and the lipid BMP 18:1_22:6 in the muscle. Our findings elucidate the complexity of molecular mechanisms linking anthropogenic and environmental stressors with vulnerability and resilience in a marine sentinel species. Furthermore, this study highlights the potential of integrative omics to define disease-related marker panels, thereby supporting ongoing and future health monitoring and conservation efforts.