Addressing antibody validation failures: a multi-stakeholder Delphi consensus study on actionable solutions
Addressing antibody validation failures: a multi-stakeholder Delphi consensus study on actionable solutions
Blades, K.; Biddle, M.; Froud, R.; Krockow, E. M.; Virk, H.
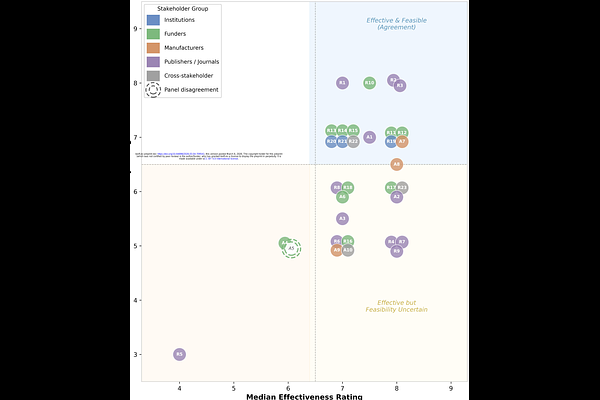
AbstractThe experimental use of antibodies that have not been validated for context-specific use frequently misdirects biomedical research. Experimental results that derive from the use of inadequately validated antibodies are estimated to waste over $1 billion annually in the United States alone and to consume millions of animal and human biological samples in experiments whose conclusions may be unreliable. Community validation frameworks, reporting standards, and independent characterisation initiatives have made important progress, and multi-stakeholder coordination efforts are emerging. However, the research community lacks a formally developed, consensus-based action plan that specifies what each stakeholder group should do, by when, and with what priority. We conducted a modified Delphi study with international experts representing academic researchers, scientific publishers, research funders, antibody manufacturers, and institutional research leaders to develop actionable recommendations for improving antibody validation, selection, and reporting practices. Thirty-two participants rated 33 proposed actions on effectiveness and feasibility using 9-point scales, with consensus assessed using the RAND/UCLA Appropriateness Method. Over two rounds, the panel achieved consensus on 15 items as both effective and feasible for implementation by 2030. These spanned institutional actions (training in antibody validation, integration into research integrity frameworks, support for local expertise networks), funder actions (dedicated validation budgets, grant application requirements, endorsement of community reporting standards), publisher actions (complete antibody reporting packages, clear validation standards), manufacturer actions (assignment of unique identifiers at source), and cross-stakeholder coordination (a shared roadmap for improvement). An additional 15 items were rated as effective but with uncertain feasibility, reflecting a consistent pattern in which the panel agreed on the value of proposed interventions but expressed reservations about realistic implementation timelines. One item was rejected by the panel with concerns around effectiveness and feasibility. Participants described four interconnected barriers to progress: diffuse ownership of the problem across stakeholders; market dynamics that inadequately reward antibody quality; difficulty justifying investment when returns are distributed across the research system; and coordination challenges among actors with different incentive structures. These barriers are addressable through coordinated action, and the findings complement existing technical and data-sharing initiatives by providing the structured, stakeholder-endorsed policy framework needed to translate awareness of the problem into concrete practice and policy changes.