Patchouli alcohol suppresses gastric cancer growth and immune evasion via inhibition of the NF-κB/PD-L1 axis
Patchouli alcohol suppresses gastric cancer growth and immune evasion via inhibition of the NF-κB/PD-L1 axis
Hou, K.; Hao, Q.; Yang, H.; Dai, F.; Wang, X.; Dai, Y. w.; Feng, L.; Lu, H.; Wang, Z.
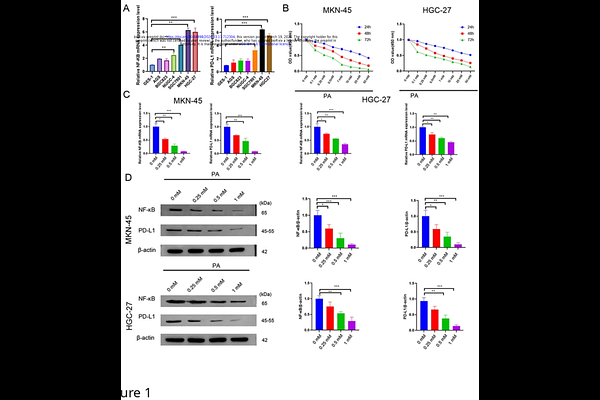
AbstractObjective This study aimed to investigate the anti-gastric cancer effect of Patchouli alcohol (PA), especially its influence on PD-L1-mediated immune evasion, and to elucidate the underlying molecular mechanisms. Methods A CCK-8 assay was used to evaluate the effects of PA on the viability of the gastric cancer cell lines HGC-27 and MKN-45. RT?qPCR and western blotting were performed to analyze the mRNA and protein levels of NF-{kappa}B and PD-L1, respectively. In a coculture system of gastric cancer cells and peripheral blood mononuclear cells (PBMCs), the effect of PA pretreatment on the PBMC-induced apoptosis of cancer cells was analyzed by flow cytometry, and the cytotoxic activity of the PBMCs was assessed by a lactate dehydrogenase (LDH) release assay. Flow cytometry was also used to determine the proportions of CD3+CD8+ T cells and IFN-{gamma}+CD8+ T cells. ELISA was used to measure the levels of IFN-{gamma}, TNF-, and granzyme B in the coculture supernatants. Immunofluorescence staining was conducted to assess NF-{kappa}B nuclear translocation. In a mouse xenograft model, tumor volume and weight were measured after 14 days of PA treatment. Histopathological changes and apoptosis were analyzed by HE and TUNEL staining. A luciferase reporter assay was used to examine the transcriptional regulation of PD-L1 by NF-{kappa}B. Results PA inhibited the viability of HGC-27 and MKN-45 cells in a dose- and time-dependent manner and significantly downregulated the expression of NF-{kappa}B and PD-L1 at both the mRNA and protein levels. In a PBMC coculture model, PA pretreatment enhanced the ability of PBMCs to induce apoptosis and directly kill gastric cancer cells. Furthermore, PA pretreatment increased the proportions of CD3+CD8+ T cells and IFN-{gamma}+CD8+ T cells in a dose-dependent manner. Consistent with this immunostimulatory effect, PA increased the levels of IFN-{gamma}, TNF-, and granzyme B in the coculture supernatants. Mechanistically, western blotting analysis demonstrated that PA significantly reduced the protein levels of AKT, NF-{kappa}B, and PD-L1 in gastric cancer cells. Immunofluorescence staining further indicated that PA suppressed the nuclear translocation of NF-{kappa}B. In a mouse xenograft model, PA treatment significantly inhibited tumor growth, induced apoptosis, and downregulated NF-{kappa}B and PD-L1 protein expression in tumor tissues. Flow cytometry of tumor-infiltrating lymphocytes revealed increased proportions of CD3+CD8+ and IFN-{gamma}+CD8+ T cells following PA treatment. Finally, luciferase reporter assays demonstrated that NF-{kappa}B directly regulates PD-L1 transcription by binding to its promoter region. Conclusion PA exerts antitumor effects in gastric cancer by suppressing the NF-{kappa}B/PD-L1 axis, thereby enhancing CD8+ T-cell-mediated cytotoxicity and inhibiting immune evasion.