A conserved isoleucine gates the diffusion of small ligands to the active site of NiFe CO-dehydrogenase
A conserved isoleucine gates the diffusion of small ligands to the active site of NiFe CO-dehydrogenase
Opdam, L.; Meneghello, M.; Guendon, C.; Chargelegue, J.; Fasano, A.; Jacq-Bailly, A.; Leger, C.; Fourmond, V.
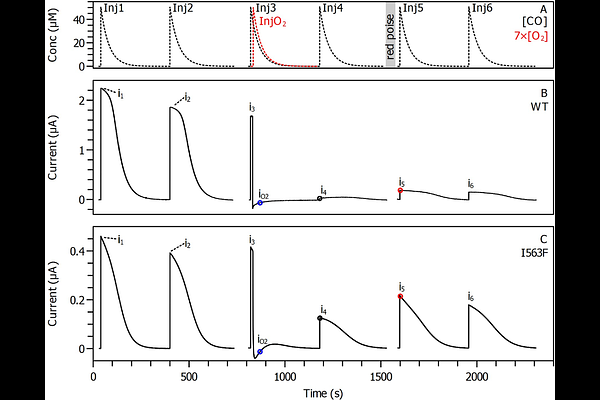
AbstractCO dehydrogenases (CODH) are metalloenzymes that reversibly oxidize CO to CO2, at a buried NiFe4S4 active site. The substrates, CO and CO2, need therefore to be transported through the protein matrix to reach the active site. The most likely pathway for intra-protein diffusion is the hydrophobic channel identified in the crystal structures. Here, we use site-directed mutagenesis to study the highly conserved isoleucine 563 of Thermococcus sp. AM4 CODH2. Mutations at this position change the biochemical properties (KM for CO, product inhibition constant, catalytic bias...), and increase the resistance of the enzyme to the inhibitor O2, showing that isoleucine 563 indeed lines the gas channel. The I563F mutation decreases the bimolecular rate constant of inhibition by O2 15-fold, and increases the IC50 20-fold, which is the strongest improvement in O2 resistance reported so far. We show that the size of the introduced amino acids is less important than their flexibility, along with the size of the cavity formed near the active site in the channel. We also conclude that O2 access to the active site cannot be slowed down without also affecting CO diffusion. This tradeoff will have to be considered in further attempts to use site-directed mutagenesis to make CODHs more O2 tolerant.