Structure-Based and Stability-Validated Prioritization of BACE1 Inhibitors Integrating Meta-Ensemble QSAR and Molecular Dynamics
Structure-Based and Stability-Validated Prioritization of BACE1 Inhibitors Integrating Meta-Ensemble QSAR and Molecular Dynamics
Chowdhury, T. D.; Shafoyat, M. U.; Hemel, N. H.; Nizam, D.; Sajib, J. H.; Toha, T. I.; Nyeem, T. A.; Farzana, M.; Haque, S. R.; Hasan, M.; Siddiquee, K. N. e. A.; Mannoor, K.
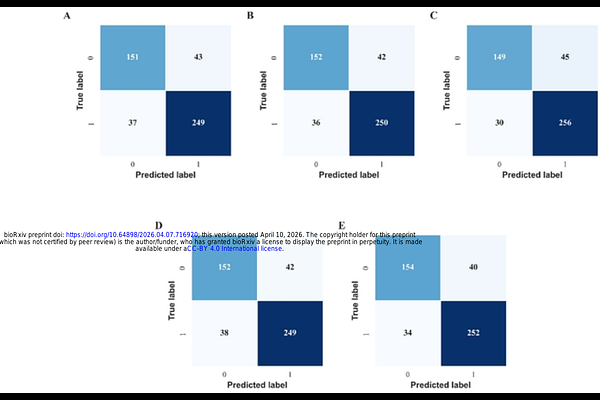
AbstractAlzheimers disease remains an unmet therapeutic challenge, and no {beta}-secretase (BACE1) inhibitor has achieved clinical approval. A major limitation of prior discovery efforts is reliance on single-parameter optimization, often yielding computational hits with poor translational potential. Here, we present a stability-validated, biology-informed computational framework that integrates meta-ensemble QSAR (five tree-based classifiers with ECFP4 fingerprints), structure-based docking, Protein Language Model (ESM-1b)-guided hybrid residue interaction weighting, and comprehensive ADMET profiling within a normalized composite ranking scheme. Model robustness was confirmed through external validation and Y-randomization (n = 100; empirical p = 0.009). Heuristic weighting was quantitatively stress-tested using global {+/-}10% perturbation analysis (mean Spearman {rho} = 0.998; mean Kendalls {tau} = 0.970), demonstrating exceptional ranking stability under controlled parameter uncertainty. Screening of 16,196 structurally diverse compounds, including CNS-active molecules, phytochemicals, approved drugs, and investigational agents, identified 153 predicted actives (accuracy 0.852; ROC-AUC 0.920), which were refined to 111 drug-like candidates and seven prioritized leads. Two-hundred-nanosecond molecular dynamics simulations confirmed stable binding within the BACE1 catalytic pocket and sustained interaction networks over time. Mol-2 exhibited the most favorable profile, characterized by low ligand RMSD (1.2-1.6 [A]), persistent catalytic dyad interactions (ASP32 98%, ASP228 99%), predicted BBB permeability, acceptable efflux profile, and balanced ADMET characteristics consistent with CNS drug-like space. Collectively, this integrative, interpretable, and robustness-validated framework provides a systematic strategy for multi-criteria lead prioritization and may serve as a transferable platform for structure-guided discovery of therapeutics targeting complex neurodegenerative pathways