Integrating Fas-mediated apoptosis with IFNγ signaling to drive tumor regression in mRNA cancer therapeutics
Integrating Fas-mediated apoptosis with IFNγ signaling to drive tumor regression in mRNA cancer therapeutics
Shin, H.-s.; Kwon, S.-G.; Lee, H.; Lee, J.-O.
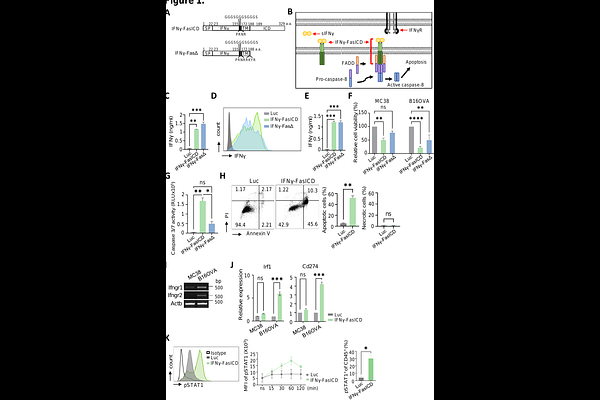
AbstractFor mRNA-based cancer gene therapy, we engineered a membrane-bound fusion protein combining interferon-gamma (IFN-gamma) with the Fas intracellular domain (FasICD) to couple local IFN-gamma signaling with Fas-driven apoptotic tumor cell death. IFN-gamma-FasICD was robustly expressed on the plasma membrane after mRNA transfection. In murine cancer cell lines, IFN-gamma-FasICD mRNA reduced viability within 24 h, resulting in ~50% cell death in MC38 cells and ~75% in B16OVA cells, exceeding the cytotoxicity of the FasICD-deleted control (IFN-gamma-Fas-deletion). Mechanistically, IFN-gamma-FasICD induced predominantly apoptotic rather than necrotic cell death. IFN-gamma-FasICD also activated IFN-gamma receptor signaling in both cancer and the immune cells, inducing IFN-gamma-responsive genes in IFN-gammaR-high B16OVA cells and triggering STAT1 phosphorylation in co-cultured splenocytes. For in vivo delivery, IFN-gamma-FasICD mRNA was formulated in lipid nanoparticles (LNPs), enabling strong intratumoral expression that peaked at ~3 h and persisted for more than 48 h. Repeated intratumoral injections of LNP-formulated IFN-gamma-FasICD mRNA suppressed the growth of established B16OVA and MC38 tumors and improved survival, with ~40% and ~20% of mice surviving beyond 30 days, respectively. IFN-gamma-FasICD treatment remodeled the tumor microenvironment by increasing tumor-infiltrating CD45+ cells and CD8+ T cells, while further reducing FOXP3+ regulatory T cells. Moreover, NK/NKT cells and cDC1/cDC2 populations were increased, and their activation was enhanced. In tumor-draining lymph nodes, IFN-gamma-FasICD mRNA promoted dendritic cell migration and increased priming and differentiation of CD8+ T cells toward effector and memory phenotypes, accompanied by enhanced functional activation of IFN-gamma-producing CD8+ T cells and highly cytotoxic NK cells in peripheral blood. Overall, our findings provide a mechanistic foundation for cytokine-death receptor fusion proteins as an in vivo antitumor strategy that can reprogram tumor cells into localized sources of both apoptotic signals and immune-activating cues.