Molecular Architecture of Cryptococcus Cell Walls Reveals Species-Specific Chitosan-Dependent Remodeling
Molecular Architecture of Cryptococcus Cell Walls Reveals Species-Specific Chitosan-Dependent Remodeling
Ankur, A.; Upadhya, R.; Doosti, M.; Ferreira, D.; Xie, L.; Hung, I.; Lodge, J.; Wang, T.
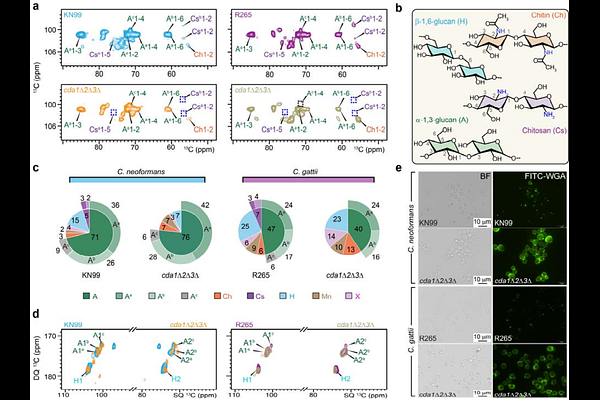
AbstractCryptococcus neoformans and Cryptococcus gattii are fungal pathogens that cause life-threatening infections, including cryptococcal meningitis. A distinctive feature of the cryptococcal cell wall is the extensive deacetylation of chitin to chitosan, a modification that is essential for virulence but whose structural role in cell-wall organization remains poorly understood. Here, we analyzed the cell walls of wild-type strains of both species and their avirulent chitosan-deficient mutants, which serve as vaccine candidates. Loss of chitosan disrupted cell morphology and altered cell-wall ultrastructure, with more pronounced defects in C. neoformans. Solid-state NMR revealed that aggregated -1,3-glucans form the principal rigid domain of the cell wall in both species and are closely associated with chitin microfibrils, whereas surrounding {beta}-glucans and mannoproteins constitute a more dynamic matrix. Chitosan modulates hydration and flexibility, and its loss increases chitin exposure and triggers species-specific remodeling of the polysaccharide network. In C. neoformans, chitosan depletion increased -1,3-glucan content and reduced {beta}-glucan levels, whereas C. gattii selectively lost one -1,3-glucan subtype while maintaining {beta}-glucan levels. Although capsule production remained intact, chitosan deficiency altered glucuronoxylomannan linkage patterns and mannoprotein composition. These findings reveal how chitosan organizes cryptococcal cell-wall architecture and highlight distinct structural adaptation strategies among pathogenic Cryptococcus species.