RUNX1-deficiency drives immune-active ER+ mammary tumorigenesis through activation of interferon signaling
RUNX1-deficiency drives immune-active ER+ mammary tumorigenesis through activation of interferon signaling
Han, S.; Xiang, D.; Chen, X.; Zhao, D.; Qin, G.; Bronson, R.; Li, Z.
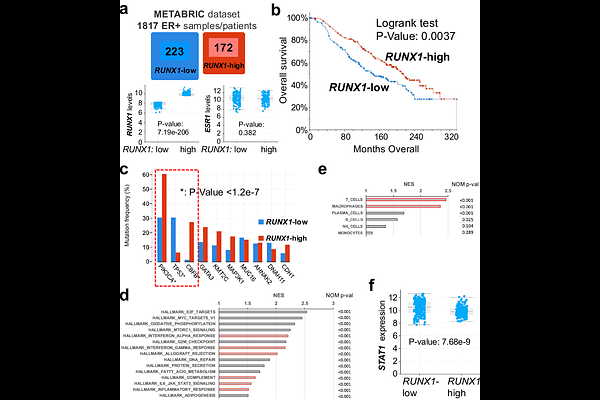
AbstractRecurrent loss-of-function mutations in RUNX1 occur in estrogen receptor-positive (ER+) breast cancers, yet how RUNX1-loss contributes to breast tumorigenesis remains unclear. Here we used genetically engineered mouse models with luminal mammary epithelial cell (MEC)-restricted gene disruption to investigate its role in breast cancer initiation. Loss of RUNX1 alone, or together with RB1, was insufficient to drive tumor formation. In contrast, combined loss of RUNX1 and p53 induced mammary tumors with full penetrance. These tumors contained ER+ cancer cells and exhibited extensive T cell and macrophage infiltration, indicative of an immune hot microenvironment. Mechanistically, RUNX1-deficiency activated interferon signaling in luminal MECs, associated with derepression of RUNX1 target STAT1 and enhanced inflammatory responses. Consistent with these findings, human ER+ breast cancers with low RUNX1 expression displayed elevated immune signatures and poorer patient survival. Together, our results identify RUNX1-loss as a driver of an immune-active subtype of ER+ breast cancer.