Disruption of RBM20 causes atrial electrophysiological disturbances
Disruption of RBM20 causes atrial electrophysiological disturbances
Weirauch, L.; Wiedmann, F.; Schraft, L.; van den Hoogenhof, M. M. G.; Prueser, M.; Kraft, M.; Wang, Y.; Paasche, A.; Dobreva, G.; Steinmetz, L.; Schmidt, C.
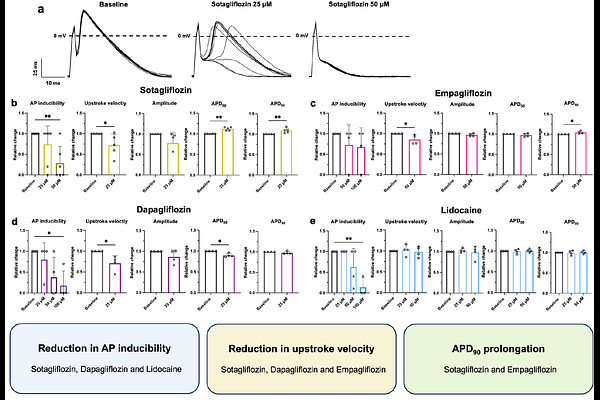
AbstractBackground Dilated cardiomyopathy (DCM) is a leading cause of heart failure, with 30 to 50 % of cases attributed to familial inheritance. Mutations in RNA-binding motif protein 20 (RBM20) account for 3 to 5 % of cases and are associated with severe DCM and ventricular arrhythmias. However, the role of RBM20 mutations in atrial cardiomyopathy (AtCM) and atrial fibrillation (AF) remains underexplored. This study investigates the effects of the RBM20-R636Q mutation on atrial electrophysiology and evaluates sodium-glucose co-transporter (SGLT) inhibitors as potential therapeutics. Results Rbm20-R636Q mice exhibited atrial remodeling, including hypertrophy, left atrial enlargement, and shortened action potential duration at 90% repolarization (APD90). Compared with RBM20-knockout and laminopathy models, RBM20-R636Q mice showed distinct reductions in Ito / IKur without changes in IK,sus or IK,tail currents, alongside TASK-1 potassium current upregulation and alterations of ICaL. SGLT inhibitors (sotagliflozin, empagliflozin, dapagliflozin) reduced AP inducibility and partially restored APD90, with effects comparable to lidocaine, suggesting a role in modulating peak sodium currents. Conclusions RBM20 mutations contribute to atrial remodeling, promoting AtCM and AF. SGLT inhibitors demonstrate therapeutic potential by modulating atrial electrophysiology and reducing arrhythmogenesis, offering a promising strategy for managing RBM20-related cardiac disorders.