A Real Time Quaking Induced Conversion (RT-QuIC) Assay for Detection of Misfolded Insulin Protein
A Real Time Quaking Induced Conversion (RT-QuIC) Assay for Detection of Misfolded Insulin Protein
Alderiso, J. M.; Hernandez LaTorre, R.; Cox, T. M.; DiGiovanni, M. G.; Fullbright, K.; Canine, B.
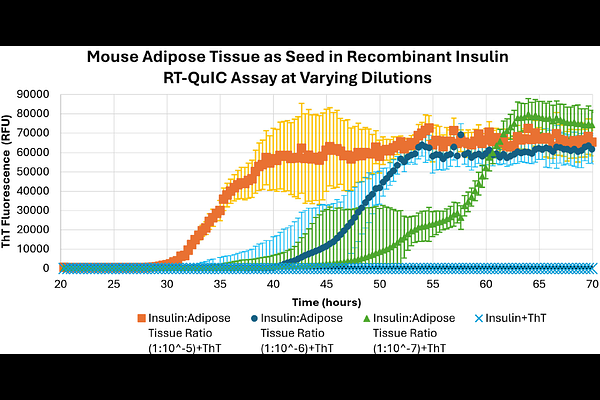
AbstractProtein misfolding plays a critical role in aging and disease, yet the involvement of specif-ic proteins in metabolic dysfunction is still poorly understood. Here, we report studies on the development of a Real-time Quaking-Induced Conversion (RT-QuIC) assay to detect misfolded insulin, a peptide hormone required for blood glucose regulation. Although RT-QuIC assays were originally designed to amplify misfolded prion proteins implicated in neurodegeneration, we adapted the method to monitor conformational changes in in-sulin. We first validated the RT-QuIC insulin assay using recombinant insulin and insu-lin aggregates recovered from clinical infusion devices. Protein characterization by gel electrophoresis, circular dichroism, and particle size analysis suggests differences in in-sulin recovered from the infusion device. We then applied the RT-QuIC assay to tissue samples from a mouse model of metabolic disease. This work provides proof-of-concept of a novel assay for studying the role of insulin aggregation in disease progression and ag-ing. The RT-QuIC assay for insulin may also provide new avenues to explore early detec-tion, mechanistic insights, and therapeutic targets of metabolic disorders linked to aging and disease.