Density-guided AlphaFold3 uncovers unmodelled conformations in β2-microglobulin
Density-guided AlphaFold3 uncovers unmodelled conformations in β2-microglobulin
Maddipatla, S. A.; Vedula, S.; Bronstein, A. M.; Marx, A.
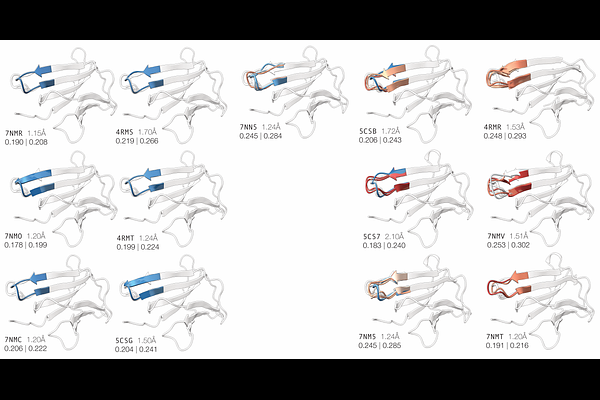
AbstractAlthough X-ray crystallography captures the ensemble of conformations present within the crystal lattice, models typically depict only the most dominant conformation, obscuring the existence of alternative states. Applying the electron density-guided AlphaFold3 approach to {beta}2-Microglobulin highlights how ensembles of alternate backbone conformations can be systematically modeled directly from crystallographic maps. This study also highlights how the detection of conformational ensembles is affected by the local quality of electron density and subtle variations in crystallization conditions and lattice packing. These results demonstrate that density-guided AlphaFold3 can uncover conformational heterogeneity missed by conventional refinement, offering a robust, systematic framework to capture the full structural landscape of proteins in crystals and enhancing the interpretive power of macromolecular crystallography.