Interactions between ploidy and resource availability shape clonal interference at initiation and recurrence of glioblastoma
Interactions between ploidy and resource availability shape clonal interference at initiation and recurrence of glioblastoma
Nowicka, Z.; Rentzeperis, F.; Beck, R.; Tagal, V.; Forero Pinto, A. M.; Scanu, E.; Veith, T.; Cole, J.; Ilter, D.; Dominguez Viqueira, W.; Teer, J. K.; Maksin, K.; Pasetto, S.; Abdalah, M. A.; Fiandaca, G.; Prabhakaran, S.; Schultz, A.; Ojwang, M.; Barnholtz-Sloan, J. S.; Joaquim, F. M.; Gomes, A. P.; Katira, P.; Andor, N.
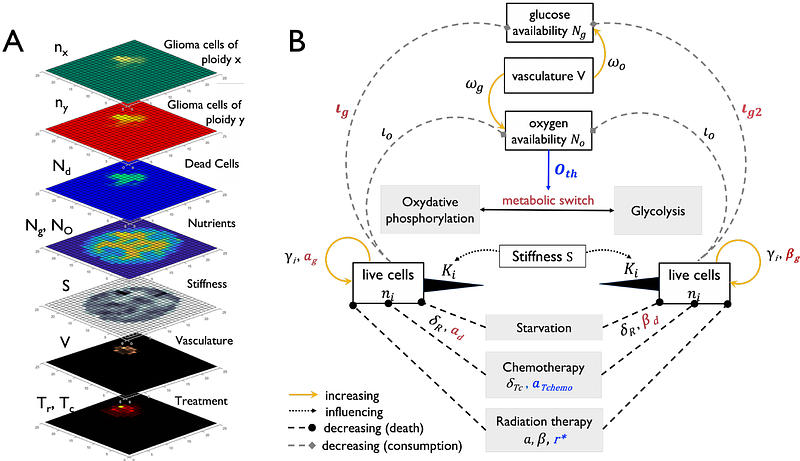
AbstractGlioblastoma (GBM) is the most aggressive form of primary brain tumor. Complete surgical resection of GBM is almost impossible due to the infiltrative nature of the cancer. While no evidence for recent selection events have been found after diagnosis, the selective forces that govern gliomagenesis are strong, shaping the tumor\'s cell composition during the initial progression to malignancy with late consequences for invasiveness and therapy response. We present a mathematical model that simulates the growth and invasion of a glioma, given its ploidy level and the nature of its brain tissue micro-environment (TME), and use it to make inferences about GBM initiation and response to standard-of-care treatment. We approximate the spatial distribution of resource access in the TME through integration of in-silico modelling, multi-omics data and image analysis of primary and recurrent GBM. In the pre-malignant setting, our in-silico results suggest that low ploidy cancer cells are more resistant to starvation-induced cell death. In the malignant setting, between first and second surgery, simulated tumors with different ploidy compositions progressed at different rates. Whether higher ploidy predicted fast recurrence, however, depended on the TME. Historical data supports this dependence on TME resources, as shown by a significant correlation between the median glucose uptake rates in human tissues and the median ploidy of cancer types that arise in the respective tissues (Spearman r = -0.70; P = 0.026). Taken together our findings suggest that availability of metabolic substrates in the TME drives different cell fate decisions for cancer cells with different ploidy and shapes GBM disease initiation and relapse characteristics.