Discovery and Biosynthesis of Nitrilobacillins by Post-translational Introduction of C-Terminal Nitrile Groups
Discovery and Biosynthesis of Nitrilobacillins by Post-translational Introduction of C-Terminal Nitrile Groups
Cha, L.; Qian, C.; Padhi, C.; Zhu, L.; van der Donk, W.
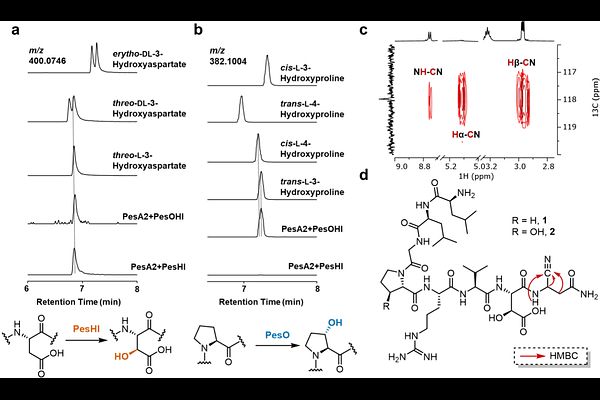
AbstractNitrile-containing natural products are produced in all kingdoms of life. Despite the wide application of nitrile-containing peptide scaffolds in medicinal chemistry, the presence of the nitrile group is unprecedented in ribosomally synthesized and post-translationally modified peptides (RiPPs). In this work, we report the identification and characterization of a RiPP biosynthetic gene cluster (BGC), where an asparagine synthetase-like (AS-like) protein encoded in the BGC converts the C-terminal carboxylate of the precursor peptide to a nitrile. Furthermore, a multinuclear nonheme iron-dependent oxidative enzyme (MNIO) and an -ketoglutarate-dependent HExxH motif-containing enzyme (KG-HExxH) perform stereoselective {beta}-hydroxylation of aspartate and proline residues, respectively. The final product is a cysteine protease inhibitor and shows that Nature makes similar warheads as found in synthetic therapeutics such as the active ingredient of Paxlovid. These findings extend our understanding of the structural and functional diversity of RiPPs.