Inducible degradation-coupled phosphoproteomics identifies PP2A Rts1 as a novel eisosome regulator
Inducible degradation-coupled phosphoproteomics identifies PP2A Rts1 as a novel eisosome regulator
DeMarco, A. G.; Dibble, M. G.; Hall, M. C.
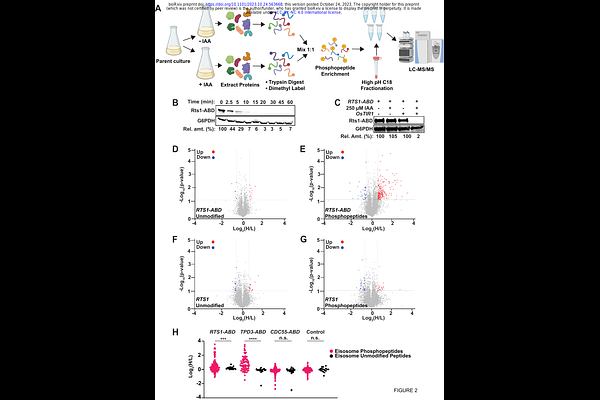
AbstractReversible protein phosphorylation is an abundant post-translational modification dynamically regulated by opposing kinases and phosphatases. Protein phosphorylation has been extensively studied in cell division, where waves of cyclin-dependent kinase activity, peaking in mitosis, drive the sequential stages of the cell cycle. Here we developed and employed a strategy to specifically probe kinase or phosphatase substrates at desired times or experimental conditions in the model organism Saccharomyces cerevisiae. We combined auxin-inducible degradation (AID) with mass spectrometry-based phosphoproteomics, which allowed us to arrest physiologically normal cultures in mitosis prior to rapid phosphatase degradation and phosphoproteome analysis. Our results revealed that protein phosphatase 2A coupled with its B56 regulatory subunit, Rts1 (PP2A Rts1), is involved in dephosphorylation of numerous proteins in mitosis, highlighting the need for phosphatases to selectively maintain certain proteins in a hypophosphorylated state in the face of high mitotic kinase activity. Unexpectedly, we observed elevated phosphorylation at many sites on several subunits of the fungal eisosome complex following rapid Rts1 degradation. Eisosomes are dynamic polymeric assemblies that create furrows in the plasma membrane important in regulating nutrient import, lipid metabolism, and stress responses, among other things. We found that PP2A Rts1-mediated dephosphorylation of eisosomes promotes their plasma membrane association and we provide evidence that this regulation impacts eisosome roles in metabolic homeostasis. The combination of rapid, inducible protein degradation with proteomic profiling offers several advantages over common protein disruption methods for characterizing substrates of regulatory enzymes involved in dynamic biological processes.